Login
Welcome back! Please enter your details.
or
Don't have an account? Register here
Create Account
Join MedMentorEdu and start your medical journey.
or
Already have an account? Login here
Enhance your knowledge with our comprehensive guide and curated study materials.
Definition
Coordinated biological response of the body against foreign antigens.
Types
Innate immunity
Present from birth
Non-specific
No immunological memory
Adaptive (acquired) immunity
Antigen-specific
Mediated by lymphocytes
Shows immunological memory
Phases of immune response
Recognition phase
Antigen recognized by BCR or TCR
Activation phase
Clonal expansion
Cytokine secretion
Effector phase
Antigen elimination
Memory phase
Formation of memory cells
Primary vs secondary immune response
Primary
Lag phase present
Low antibody titre
Predominantly IgM
Secondary
No lag phase
Rapid and intense response
Predominantly IgG

Humoral effector mechanisms
Neutralization of toxins and viruses
Opsonization → enhanced phagocytosis
Complement activation
Cell-mediated effector mechanisms
Cytotoxic T-cell mediated killing
Activated macrophage-mediated destruction
Antibody-dependent cellular cytotoxicity (ADCC)
IgG-coated target cell destroyed by NK cells
Complement-mediated lysis
Formation of membrane attack complex (MAC)

Definition
Immune response mediated by T lymphocytes without antibody involvement
Cells involved
CD4⁺ helper T cells
CD8⁺ cytotoxic T cells
Macrophages
Natural killer (NK) cells
Antigen presentation
MHC I → CD8⁺ T cells
MHC II → CD4⁺ T cells
Functions
Defense against intracellular pathogens
Tumor immunity
Graft rejection
Type IV (delayed) hypersensitivity
Key cytokines
IL-2 → T-cell proliferation
IFN-γ → macrophage activation
Examples
Tuberculin skin test
Contact dermatitis
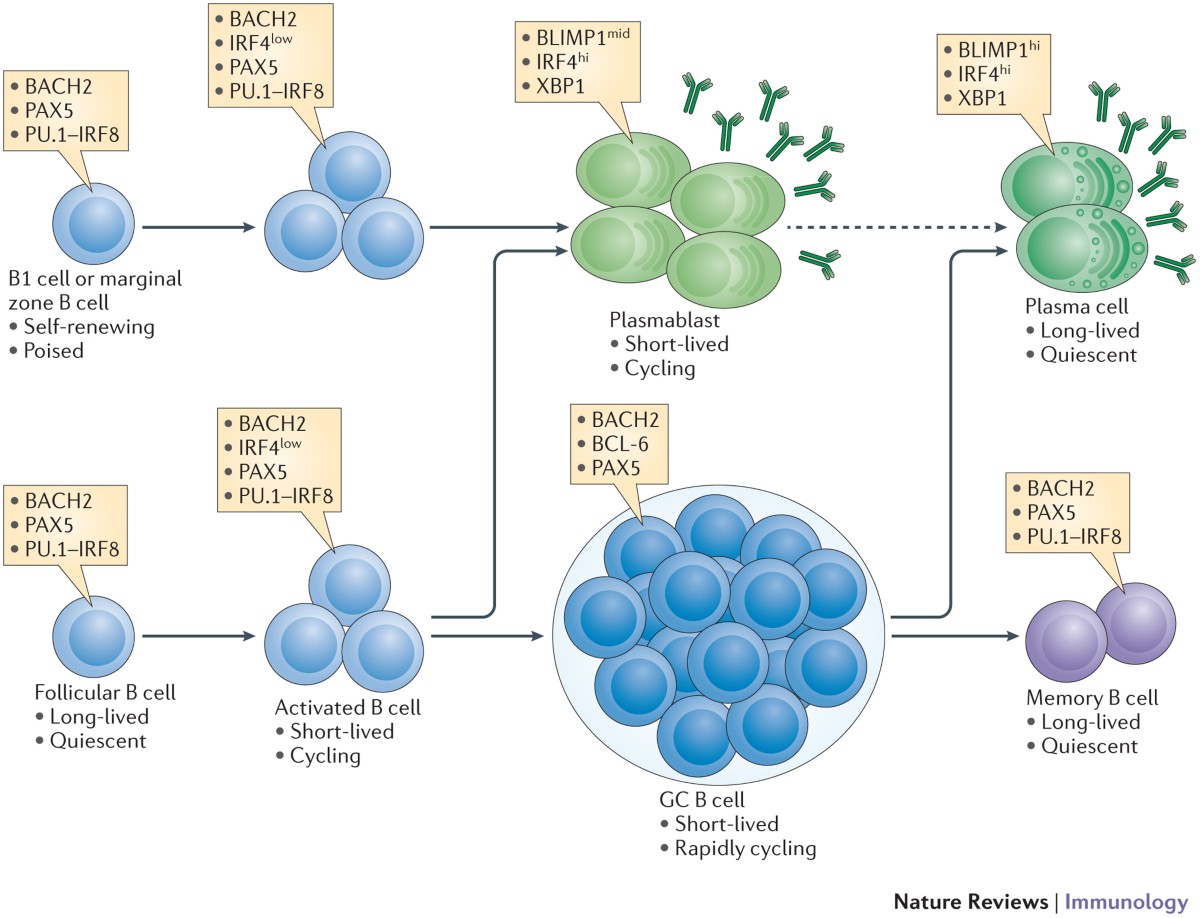
Definition
Antibody-mediated immune response
Cells involved
B lymphocytes
Plasma cells
Mechanism
Antigen binds B-cell receptor
T-dependent or T-independent activation
Differentiation into plasma cells
Functions of antibodies
Neutralization
Opsonization
Complement activation
Agglutination
Important against
Extracellular bacteria
Toxin-mediated diseases
Basic structure
Y-shaped glycoprotein molecule
Chains
2 heavy (H) chains
2 light (L) chains
Light chains
Kappa (κ)
Lambda (λ)
Heavy chains
Gamma (γ), Alpha (α), Mu (μ), Delta (δ), Epsilon (ε)
Regions
Variable region
Antigen-binding site
Determines specificity
Constant region
Determines biological function
Fragments
Fab
Antigen binding
Fc
Complement activation
Binding to Fc receptors
Hinge region
Provides flexibility
Disulfide bonds
Maintain structural stability
IgG
Most abundant
Crosses placenta
Dominant in secondary immune response
IgA
Secretory immunoglobulin
Present in saliva, tears, breast milk
IgM
First antibody produced
Pentameric structure
IgE
Involved in allergy and anaphylaxis
Defense against parasites
IgD
Acts as B-cell receptor

Definition
Ability of antibodies to recognize an enormous variety of antigens.
Basis of variability
Multiple genes for light and heavy chains
V(D)J gene recombination
V (variable)
D (diversity) – heavy chain only
J (joining)
Junctional diversity
Somatic hypermutation
Region responsible
Variable (V) region of heavy and light chains
Significance
Enables recognition of millions of different antigens
Classification based on heavy chain
γ → IgG
μ → IgM
α → IgA
ε → IgE
δ → IgD
Biological role depends on
Fc (constant) region of heavy chain
Most abundant immunoglobulin in serum
Major antibody of secondary immune response
Functions
Opsonization
Neutralization of toxins and viruses
Complement activation
Special features
Crosses placenta → passive immunity to fetus
Longest half-life
Subclasses
IgG1, IgG2, IgG3, IgG4

First antibody produced in primary immune response
Structure
Pentamer
Linked by J chain
Functions
Strong complement activation
Agglutination
Clinical importance
Marker of recent or acute infection
Mainly intravascular
Major immunoglobulin in secretions
Structure
Dimer with J chain
Secretory component protects from digestion
Found in
Saliva
Tears
Colostrum and breast milk
Function
Mucosal immunity
Prevents microbial adherence
Lowest concentration in serum
Binds to
Mast cells
Basophils
Functions
Type I hypersensitivity reactions
Defense against parasitic infections
Mechanism
Antigen–IgE interaction → mast cell degranulation → histamine release
Definition
Antigenic differences between immunoglobulin classes
Determined by
Constant region of heavy chain
Examples
IgG, IgM, IgA, IgE, IgD
Present in
All normal individuals of a species
Definition
Antigenic differences between immunoglobulins of different individuals of the same species
Basis
Genetic polymorphism
Location
Constant region of heavy or light chains
Clinical relevance
Transfusion reactions
Transplant immunology
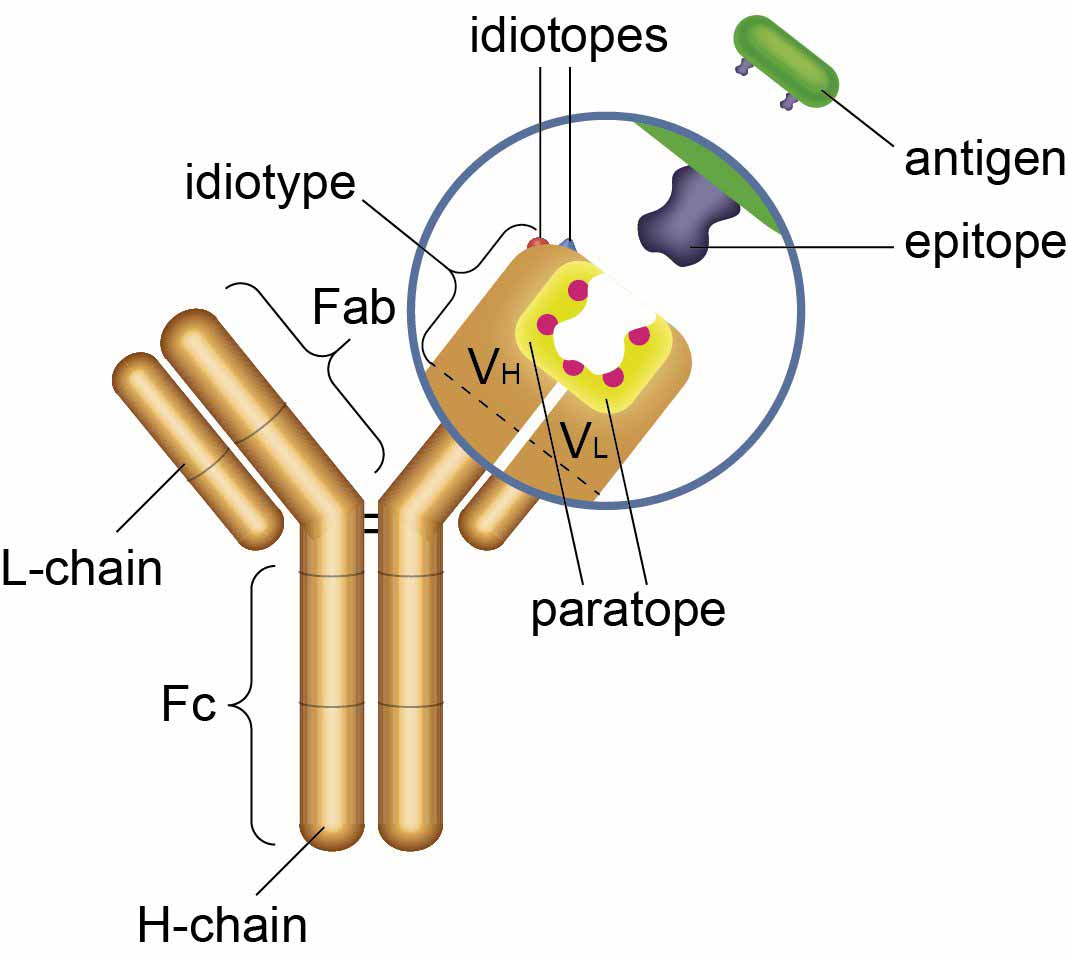

Definition
Unique antigenic determinants present in the variable region
Location
Antigen-binding site (Fab region)
Specific to
Each individual antibody
Importance
Antibody specificity
Immune regulation (anti-idiotype antibodies)



Definition
Malignant clonal proliferation of plasma cells producing a single monoclonal immunoglobulin.
Type of immunoglobulin
Most commonly IgG
IgA less common
Rarely light chains only
Pathogenesis
Plasma cell proliferation in bone marrow
Excess monoclonal protein (M protein)
Biochemical features
Monoclonal gammopathy
M spike on serum protein electrophoresis
Decreased normal immunoglobulins
Clinical features
Bone pain
Pathological fractures
Recurrent infections
Anemia
Renal involvement
Light chain deposition
Cast nephropathy
Complications
Hypercalcemia
Renal failure
Diagnosis
Bone marrow plasma cells >10%
Serum/urine monoclonal protein

Definition
Localized tumor of monoclonal plasma cells.
Types
Solitary bone plasmacytoma
Extramedullary plasmacytoma
Difference from multiple myeloma
Single lesion
No generalized bone marrow involvement
Immunoglobulin production
May produce monoclonal immunoglobulin
Clinical significance
Can progress to multiple myeloma

Definition
Presence of free immunoglobulin light chains in urine.
Seen in
Multiple myeloma
Plasma cell dyscrasias
Type of protein
Kappa or lambda light chains
Special property
Precipitates at 40–60°C
Redissolves on boiling
Reappears on cooling
Biochemical importance
Causes renal tubular damage
Leads to myeloma kidney
Diagnostic value
Marker of plasma cell malignancy
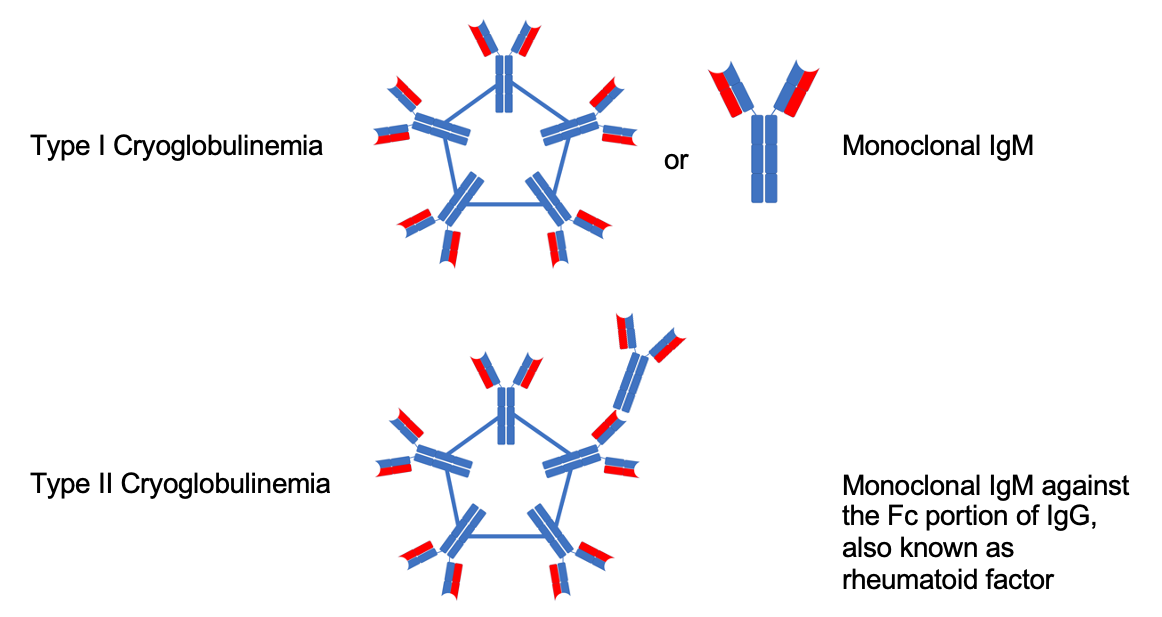
Definition
Lymphoplasmacytic malignancy producing excess IgM.
Type of immunoglobulin
IgM (macroglobulin)
Biochemical features
Very high molecular weight IgM
Increased serum viscosity
Clinical features
Hyperviscosity syndrome
Visual disturbances
Bleeding tendency
Neurological symptoms
Difference from multiple myeloma
Bone lesions usually absent
Renal damage less common

Definition
Increased gamma globulin fraction in serum.
Types
Polyclonal hypergammaglobulinemia
Monoclonal hypergammaglobulinemia
Polyclonal causes
Chronic infections
Autoimmune diseases
Liver disease
Monoclonal causes
Multiple myeloma
Macroglobulinemia
Serum protein electrophoresis
Polyclonal → broad-based gamma peak
Monoclonal → sharp M spike
Clinical significance
Marker of chronic immune stimulation or plasma cell disorder
Myeloma → monoclonal Ig + bone destruction
Plasmacytoma → localized plasma cell tumor
Bence-Jones → light chains in urine
Macroglobulinemia → IgM + hyperviscosity
Hypergammaglobulinemia → increased gamma fraction
Definition
A group of plasma proteins that enhance antigen–antibody reactions and innate immunity.
Nature
Heat-labile
Synthesized mainly by liver
Components
C1 to C9
Pathways of activation
Classical pathway
Triggered by antigen–antibody (IgG, IgM) complex
Alternative pathway
Activated directly by microbial surfaces
Lectin pathway
Activated by mannose-binding lectin
Common terminal pathway
Formation of C5b–C9 membrane attack complex (MAC)
Biological functions
Cell lysis
Opsonization (C3b)
Chemotaxis (C5a)
Inflammation
Regulation
Prevents damage to host cells
Controlled by inhibitors like C1 esterase inhibitor
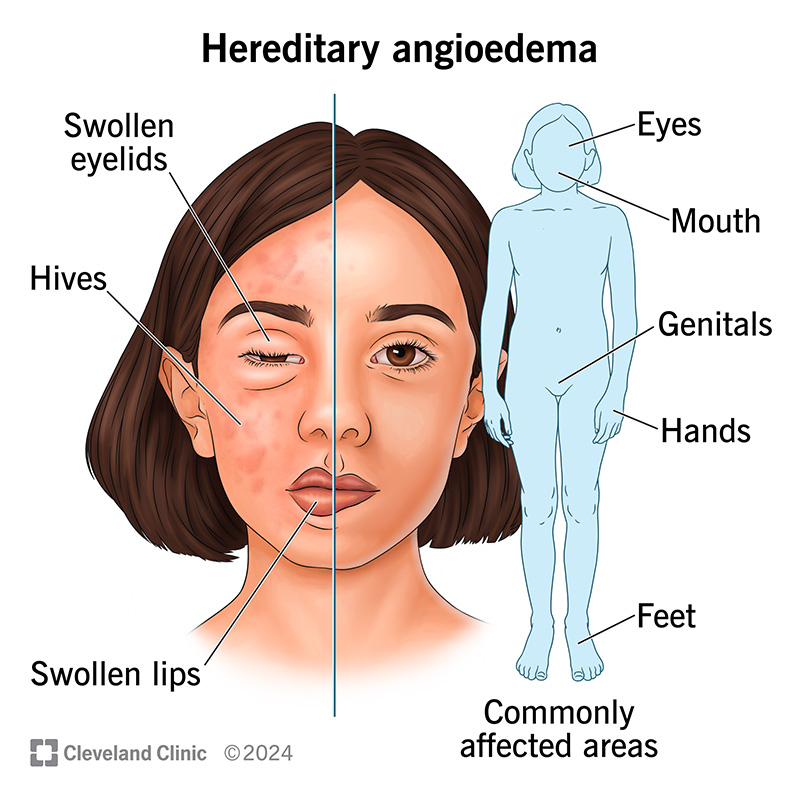
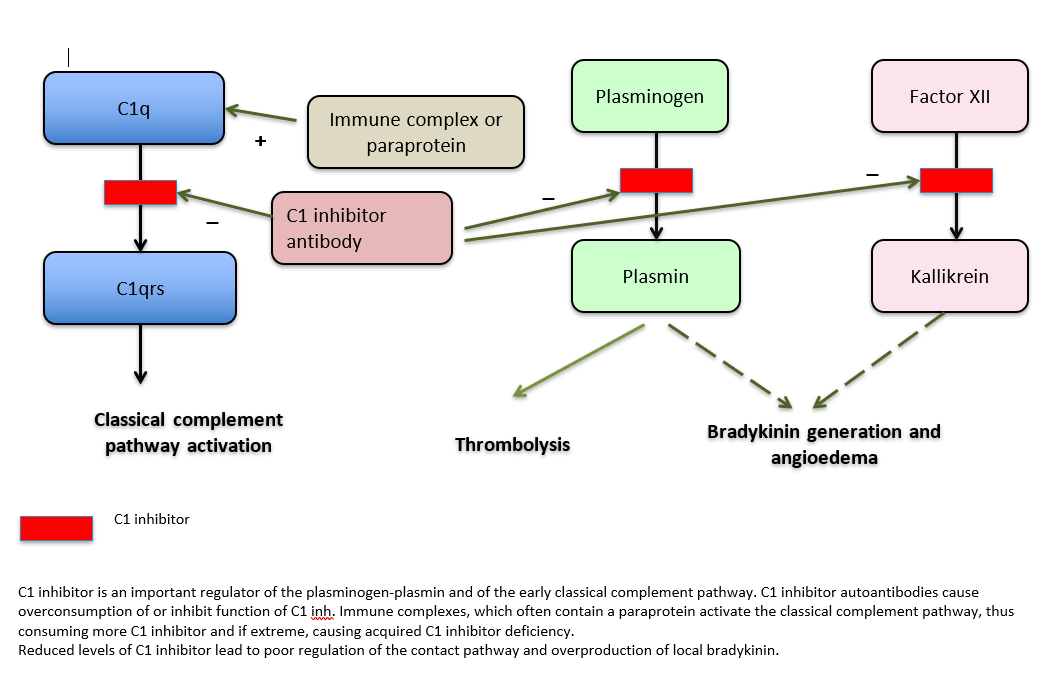
Definition
Autosomal dominant disorder due to deficiency of C1 esterase inhibitor.
Pathophysiology
Uncontrolled activation of complement system
Excess bradykinin formation
Biochemical defect
Decreased or dysfunctional C1 esterase inhibitor
Clinical features
Recurrent non-pitting edema
Involves skin, gastrointestinal tract, larynx
No urticaria or itching
Triggers
Trauma
Stress
Dental procedures
Laboratory findings
Low C4 levels
Treatment
C1 esterase inhibitor concentrate
Bradykinin receptor antagonists

Definition
Conditions where immune response is impaired or absent.
Classification
Primary immunodeficiency
Congenital
Genetic defects
Secondary immunodeficiency
Acquired
Primary immunodeficiency
B-cell defects → ↓ antibody production
T-cell defects → impaired cell-mediated immunity
Combined defects → severe infections
Secondary immunodeficiency causes
Malnutrition
HIV infection
Malignancy
Chemotherapy
Immunosuppressive drugs
Clinical features
Recurrent infections
Opportunistic infections
Poor response to vaccines



Gene organization
Separate gene segments for variable (V), diversity (D), and joining (J) regions
V(D)J recombination
Random rearrangement of gene segments
Generates antibody diversity
Junctional diversity
Addition or deletion of nucleotides at joining sites
Somatic hypermutation
Point mutations in variable region
Increases antibody affinity
Class switch recombination
Change in heavy chain constant region
IgM → IgG / IgA / IgE
Antigen specificity unchanged
Role of enzymes
RAG enzymes → gene rearrangement
Activation-induced cytidine deaminase (AID) → class switching and hypermutation
Final outcome
Production of high-affinity, antigen-specific antibodies
Complement → lysis, opsonization, inflammation
Hereditary angioedema → C1 esterase inhibitor deficiency
Immunodeficiency → recurrent infections
Antibody production → V(D)J recombination + class switching
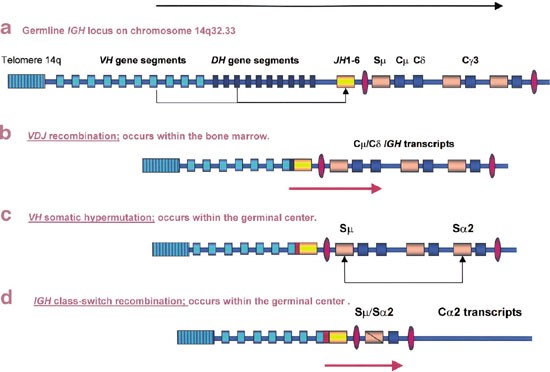

Definition
Movement and rearrangement of gene segments within the genome during lymphocyte development.
Relevance in immunity
Essential for immunoglobulin and T-cell receptor diversity.
Mechanism
Rearrangement of V (variable), D (diversity), and J (joining) gene segments.
Enzymes involved
RAG-1 and RAG-2 (recombination activating genes)
Outcome
Generation of vast antibody repertoire from limited genes
Site
Occurs in developing B and T lymphocytes
Definition
DNA rearrangement occurring in somatic (non-germline) cells of immune system.
Occurs in
B lymphocytes → immunoglobulin genes
T lymphocytes → TCR genes
Steps
Selection of V, D, J segments
Joining with deletion of intervening DNA
Significance
Each lymphocyte expresses a unique antigen receptor
Clinical relevance
Defects → severe combined immunodeficiency (SCID)
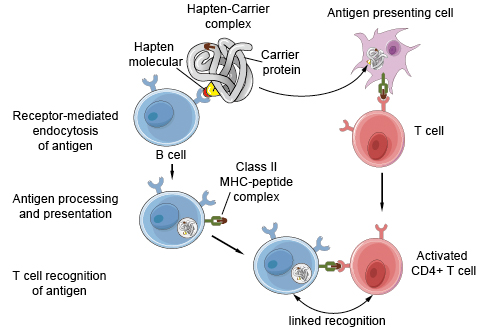
Definition
Any substance capable of inducing an immune response.
Chemical nature
Proteins (most potent)
Polysaccharides
Lipoproteins
Nucleoproteins
Antigenic determinant (Epitope)
Specific region recognized by antibody or TCR
Properties influencing antigenicity
Foreignness
Molecular size
Chemical complexity
Haptens
Small molecules
Become antigenic only when bound to carrier protein
Definition
Human leukocyte antigens involved in antigen presentation.
Location
Chromosome 6
Classes
Class I (HLA-A, B, C)
Present on all nucleated cells
Present endogenous antigens
Interact with CD8⁺ T cells
Class II (HLA-DR, DQ, DP)
Present on antigen-presenting cells
Present exogenous antigens
Interact with CD4⁺ T cells
Functions
Antigen presentation
Self–nonself discrimination
Transplant compatibility
Clinical importance
Organ transplantation
Autoimmune diseases
Disease susceptibility
Definition
Small soluble proteins that mediate communication between immune cells.
Produced by
Lymphocytes
Macrophages
Endothelial cells
Characteristics
Act at low concentrations
Bind specific receptors
Short half-life
Major types
Interleukins
Interferons
Tumor necrosis factors
Functions
Cell activation
Proliferation
Differentiation
Inflammation regulation
Definition
Cytokines produced specifically by lymphocytes.
Major sources
Activated T helper cells
Examples
IL-2 → T-cell proliferation
IFN-γ → macrophage activation
IL-4 → B-cell differentiation
Functions
Regulation of immune response
Coordination between humoral and cell-mediated immunity
Role in hypersensitivity
Especially Type IV (delayed type)
Gene transposition + somatic recombination → antibody diversity
Antigen → epitope-based recognition
HLA → antigen presentation + transplant fate
Cytokines → immune communication
Lymphokines → T-cell control signals
What is transposition of genes in the immune system?
It is the rearrangement of immunoglobulin and T-cell receptor gene segments (V, D, J) during lymphocyte development to generate antibody diversity.
Which enzymes are responsible for gene transposition in antibody formation?
Recombination activating genes RAG-1 and RAG-2.
What is somatic recombination?
DNA rearrangement occurring in somatic lymphoid cells leading to unique antigen receptors on each B or T cell.
How does somatic recombination differ from germline recombination?
Somatic recombination occurs in immune cells after birth, whereas germline recombination occurs during gamete formation and is inherited.
What is an antigen?
A substance capable of inducing an immune response and specifically reacting with antibodies or T-cell receptors.
What is an epitope?
The specific antigenic determinant on an antigen molecule that binds to an antibody or T-cell receptor.
Which chemical substances are most antigenic?
Proteins are the most potent antigens due to their large size and chemical complexity.
What is a hapten?
A small molecule that is antigenic but not immunogenic unless attached to a carrier protein.
What are HLA antigens?
Human leukocyte antigens are cell surface proteins involved in antigen presentation and immune regulation.
Where are HLA genes located?
On chromosome 6.
Differentiate HLA class I and class II.
Class I → present on all nucleated cells, present endogenous antigens, interact with CD8⁺ T cells
Class II → present on antigen-presenting cells, present exogenous antigens, interact with CD4⁺ T cells
Why are HLA antigens important clinically?
They determine transplant compatibility and are associated with autoimmune diseases.
What are cytokines?
Low-molecular-weight proteins that mediate communication between immune cells.
Name important cytokines and their functions.
IL-2 → T-cell proliferation
IFN-γ → macrophage activation
TNF-α → inflammation and apoptosis
What are lymphokines?
Cytokines produced specifically by activated lymphocytes.
How do lymphokines differ from cytokines?
Lymphokines are a subset of cytokines produced by lymphocytes only.
What is the role of cytokines in immune response?
They regulate activation, proliferation, differentiation, and coordination of immune cells.
What happens if cytokine regulation is lost?
It may lead to cytokine storm, chronic inflammation, or immunodeficiency.
How do antibodies achieve enormous diversity despite limited genes?
Through gene transposition, somatic recombination, junctional diversity, and somatic hypermutation.
Which defect leads to severe combined immunodeficiency (SCID)?
Failure of somatic recombination due to RAG gene defects.
Genes rearrange → antigens are recognized → HLA presents → cytokines communicate → immunity executes
1. Antibody diversity is primarily generated by:
A. Alternative splicing
B. Gene amplification
C. Somatic recombination
D. RNA editing
Answer: C
2. Which gene segments are involved in immunoglobulin heavy chain rearrangement?
A. V and J only
B. V, D and J
C. D and J only
D. V and C
Answer: B
3. Which enzyme initiates V(D)J recombination?
A. DNA polymerase
B. RNA polymerase
C. RAG-1 and RAG-2
D. Topoisomerase
Answer: C
4. Somatic recombination occurs in:
A. Germ cells
B. All body cells
C. Lymphoid cells
D. Hepatocytes
Answer: C
5. Failure of somatic recombination leads to:
A. Hypergammaglobulinemia
B. SCID
C. Multiple myeloma
D. Autoimmunity
Answer: B
6. The antigen-binding site of an antibody is formed by:
A. Constant region
B. Fc fragment
C. Variable region
D. Hinge region
Answer: C
7. An epitope is best defined as:
A. Entire antigen molecule
B. Antibody binding site
C. Antigenic determinant
D. Fc region
Answer: C
8. Which substance is most immunogenic?
A. Lipids
B. Proteins
C. Nucleic acids
D. Polysaccharides
Answer: B
9. Haptens become immunogenic when:
A. Heated
B. Oxidized
C. Linked to carrier proteins
D. Injected intravenously
Answer: C
10. HLA genes are located on:
A. Chromosome 2
B. Chromosome 6
C. Chromosome 11
D. Chromosome 14
Answer: B
11. HLA class I molecules present antigen to:
A. CD4⁺ T cells
B. CD8⁺ T cells
C. B cells
D. NK cells
Answer: B
12. HLA class II molecules are expressed on:
A. All nucleated cells
B. RBCs
C. Antigen-presenting cells
D. Platelets
Answer: C
13. Which HLA class presents endogenous antigens?
A. Class I
B. Class II
C. Class III
D. Beta-2 microglobulin
Answer: A
14. The most polymorphic genes in humans are:
A. Immunoglobulin genes
B. Cytokine genes
C. HLA genes
D. Complement genes
Answer: C
15. Cytokines are best described as:
A. Hormones
B. Enzymes
C. Low molecular weight signaling proteins
D. Structural proteins
Answer: C
16. Which cytokine stimulates T-cell proliferation?
A. IL-1
B. IL-2
C. IL-4
D. IL-10
Answer: B
17. IFN-γ mainly activates:
A. B cells
B. Mast cells
C. Macrophages
D. Neutrophils
Answer: C
18. Cytokines act by binding to:
A. Nuclear receptors
B. Cell surface receptors
C. DNA directly
D. Ribosomes
Answer: B
19. Lymphokines are produced mainly by:
A. Macrophages
B. Neutrophils
C. Lymphocytes
D. Endothelial cells
Answer: C
20. Which is NOT a property of cytokines?
A. Pleiotropy
B. Redundancy
C. Long half-life
D. Specific receptors
Answer: C
21. Complement proteins are primarily synthesized by:
A. Spleen
B. Bone marrow
C. Liver
D. Thymus
Answer: C
22. The classical complement pathway is activated by:
A. Bacterial endotoxin
B. IgG or IgM antigen-antibody complex
C. Mannose
D. Zymosan
Answer: B
23. The key opsonin of complement system is:
A. C1
B. C3b
C. C5a
D. C9
Answer: B
24. Membrane attack complex is composed of:
A. C1–C3
B. C3–C5
C. C5b–C9
D. C6–C8
Answer: C
25. Hereditary angioneurotic edema is due to deficiency of:
A. C3
B. C5
C. C1 esterase inhibitor
D. Factor H
Answer: C
26. Hereditary angioedema is characterized by:
A. Urticaria with itching
B. Bradykinin-mediated edema
C. Histamine release
D. Elevated IgE
Answer: B
27. Low levels of which complement component are seen in hereditary angioedema?
A. C1
B. C3
C. C4
D. C9
Answer: C
28. Primary immunodeficiency disorders are:
A. Acquired
B. Drug-induced
C. Genetic
D. Nutritional
Answer: C
29. Secondary immunodeficiency can be caused by:
A. RAG mutation
B. Malnutrition
C. Thymic aplasia
D. X-linked defects
Answer: B
30. Recurrent opportunistic infections suggest:
A. Autoimmunity
B. Immunodeficiency
C. Hypersensitivity
D. Allergy
Answer: B
31. Class switch recombination changes:
A. Antigen specificity
B. Variable region
C. Constant region of heavy chain
D. Light chain type
Answer: C
32. Enzyme essential for class switch recombination is:
A. DNA ligase
B. RNA polymerase
C. Activation-induced cytidine deaminase
D. Telomerase
Answer: C
33. Somatic hypermutation occurs in:
A. Constant region
B. Variable region
C. Fc region
D. Hinge region
Answer: B
34. Purpose of somatic hypermutation is to:
A. Increase antibody quantity
B. Increase affinity
C. Change antibody class
D. Decrease autoimmunity
Answer: B
35. Which immunoglobulin is produced first in immune response?
A. IgG
B. IgA
C. IgM
D. IgE
Answer: C
36. IgM is especially efficient in:
A. Placental transfer
B. Complement activation
C. Allergy
D. Mucosal immunity
Answer: B
37. Secretory IgA is mainly involved in:
A. Serum immunity
B. Placental immunity
C. Mucosal defense
D. Allergy
Answer: C
38. IgE binds strongly to:
A. Neutrophils
B. Mast cells
C. RBCs
D. Platelets
Answer: B
39. HLA typing is most important for:
A. Vaccine development
B. Blood transfusion
C. Organ transplantation
D. Allergy testing
Answer: C
40. Polyclonal hypergammaglobulinemia is seen in:
A. Multiple myeloma
B. Waldenström macroglobulinemia
C. Chronic infections
D. Plasma cell tumor
Answer: C
41. Monoclonal gammopathy shows which electrophoretic pattern?
A. Broad gamma band
B. Sharp M spike
C. Decreased albumin
D. No gamma globulin
Answer: B
42. Bence-Jones proteins are:
A. Heavy chains
B. Light chains
C. Complement proteins
D. Cytokines
Answer: B
43. Bence-Jones proteins are best detected in:
A. Serum
B. CSF
C. Urine
D. Saliva
Answer: C
44. Waldenström macroglobulinemia is characterized by excess:
A. IgG
B. IgA
C. IgM
D. IgE
Answer: C
45. Hyperviscosity syndrome is classically seen in:
A. Multiple myeloma
B. Plasmacytoma
C. Waldenström macroglobulinemia
D. SCID
Answer: C
46. Complement deficiency most commonly leads to:
A. Allergy
B. Autoimmunity
C. Recurrent infections
D. Cancer
Answer: C
47. Cytokines usually act in which manner?
A. Endocrine
B. Autocrine and paracrine
C. Intracrine only
D. Neurocrine
Answer: B
48. Which cytokine is pro-inflammatory?
A. IL-10
B. TGF-β
C. TNF-α
D. IL-4
Answer: C
49. Gene rearrangement in immunity is an example of:
A. Point mutation
B. Chromosomal deletion
C. Controlled DNA recombination
D. RNA splicing
Answer: C
50. The ultimate purpose of gene transposition in immunity is:
A. Faster cell division
B. Antibody class switching
C. Antigen recognition diversity
D. Immune tolerance
Answer: C
What is somatic recombination?
Rearrangement of V, D, and J gene segments in lymphocytes to generate antigen receptor diversity.
Where does somatic recombination occur?
In developing B and T lymphocytes.
Name the enzymes responsible for V(D)J recombination.
RAG-1 and RAG-2.
What is gene transposition in immunology?
Movement and rearrangement of immunoglobulin gene segments during antibody synthesis.
What is an epitope?
The specific antigenic determinant that binds to an antibody or T-cell receptor.
What is a hapten?
A small molecule that becomes immunogenic only when attached to a carrier protein.
Define HLA.
Human leukocyte antigens are MHC molecules involved in antigen presentation.
Location of HLA genes?
Chromosome 6.
Difference between HLA class I and class II?
Class I presents endogenous antigens to CD8⁺ T cells; class II presents exogenous antigens to CD4⁺ T cells.
What are cytokines?
Low-molecular-weight proteins that regulate immune cell communication.
What are lymphokines?
Cytokines produced by lymphocytes.
What is complement system?
A group of plasma proteins that enhance immune defense via lysis, opsonization, and inflammation.
Which complement component is the key opsonin?
C3b.
What is MAC?
Membrane attack complex formed by C5b–C9 causing cell lysis.
What is hereditary angioneurotic edema?
An autosomal dominant disorder due to C1 esterase inhibitor deficiency.
Why is urticaria absent in hereditary angioedema?
Edema is bradykinin-mediated, not histamine-mediated.
What is immunodeficiency?
Failure of immune system to mount an adequate response.
Difference between primary and secondary immunodeficiency?
Primary is genetic; secondary is acquired.
A 55-year-old man presents with bone pain, recurrent infections, anemia, and renal failure. Serum protein electrophoresis shows a sharp M-spike.
Diagnosis?
Multiple myeloma.
Biochemical abnormality?
Monoclonal immunoglobulin production.
Protein responsible for renal damage?
Bence-Jones proteins (light chains).
Immunoglobulin commonly increased?
IgG.
A patient has recurrent episodes of facial and laryngeal swelling without itching or urticaria. C4 levels are low.
Diagnosis?
Hereditary angioneurotic edema.
Deficient protein?
C1 esterase inhibitor.
Mediator causing edema?
Bradykinin.
A child presents with recurrent bacterial and fungal infections since infancy. Both humoral and cell-mediated immunity are defective.
Likely diagnosis?
Severe combined immunodeficiency (SCID).
Underlying defect?
Failure of somatic recombination.
Enzyme involved?
RAG gene defect.
A patient develops blurred vision, headache, and bleeding tendency. Serum shows very high IgM levels.
Diagnosis?
Waldenström macroglobulinemia.
Pathophysiology?
Hyperviscosity due to excess IgM.
Bone lesions present or absent?
Absent.
A patient with chronic liver disease shows a broad-based increase in gamma globulin region on electrophoresis.
Diagnosis?
Polyclonal hypergammaglobulinemia.
Cause?
Chronic immune stimulation.
Difference from multiple myeloma?
No monoclonal M-spike.
A newborn receives passive immunity from mother.
Which immunoglobulin crosses placenta?
IgG.
Mechanism involved?
Fc receptor-mediated transport.
A patient with parasitic infection develops marked eosinophilia and allergic manifestations.
Immunoglobulin involved?
IgE.
Cells to which IgE binds?
Mast cells and basophils.
After organ transplantation, graft rejection occurs.
Major molecules responsible?
HLA antigens.
Which class is most important in graft rejection?
HLA class I and II.
SCID → failed V(D)J recombination
Hereditary angioedema → low C4
Myeloma → monoclonal gammopathy
Waldenström → IgM + hyperviscosity
HLA → transplant fate
C3b → opsonization
Get the full PDF version of this chapter.