Topic Overview
🧠 CENTRAL NERVOUS SYSTEM
🔶 PATTERNS OF INJURY IN THE NERVOUS SYSTEM
🔹 Neuronal Injury
🔸 Reversible Injury
-
Cellular swelling (hydropic change)
-
Mild mitochondrial dysfunction
-
Decreased ATP production (early stage)
-
No nuclear changes
-
Potential for recovery if insult removed
🔸 Irreversible Injury
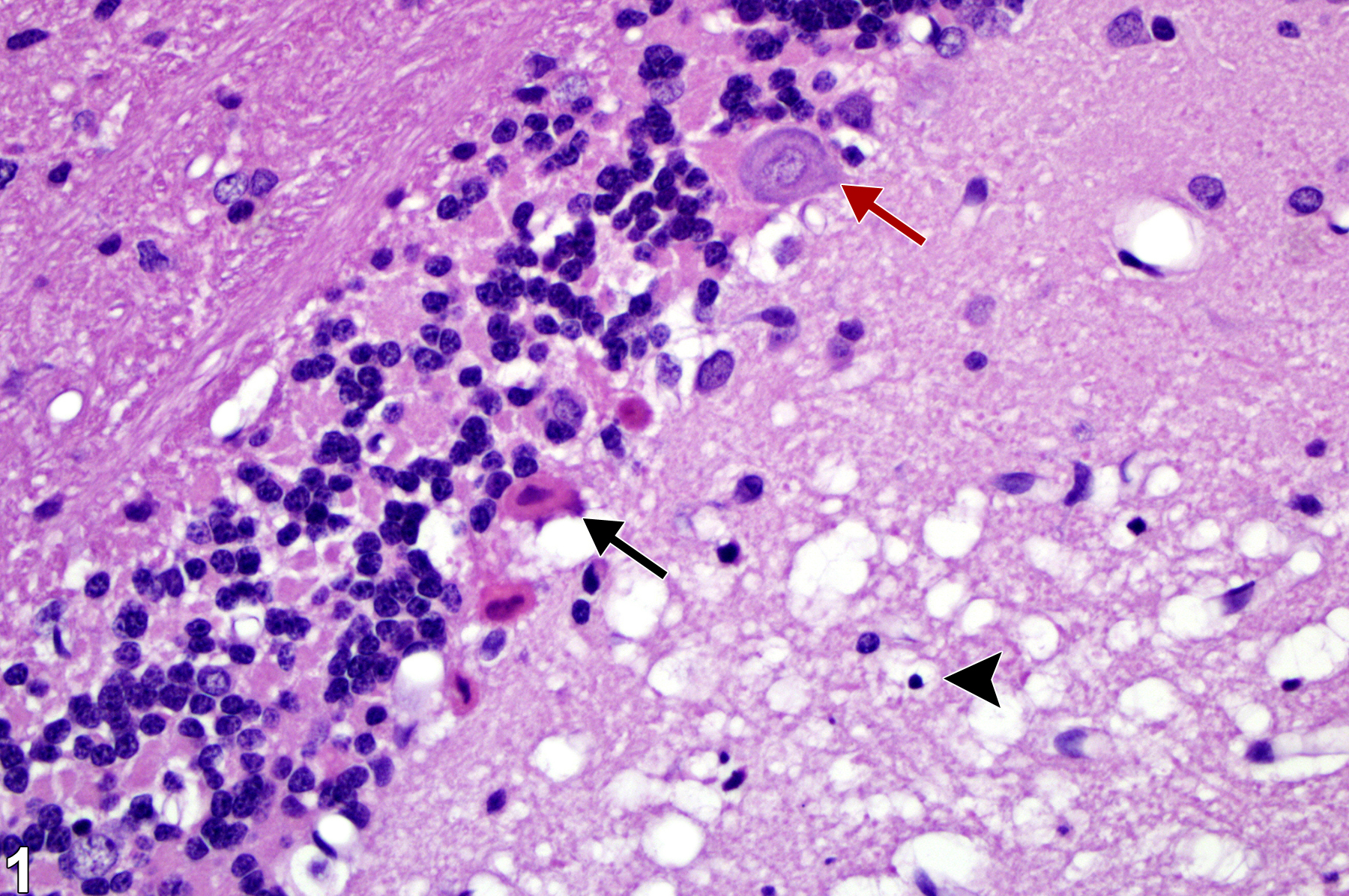
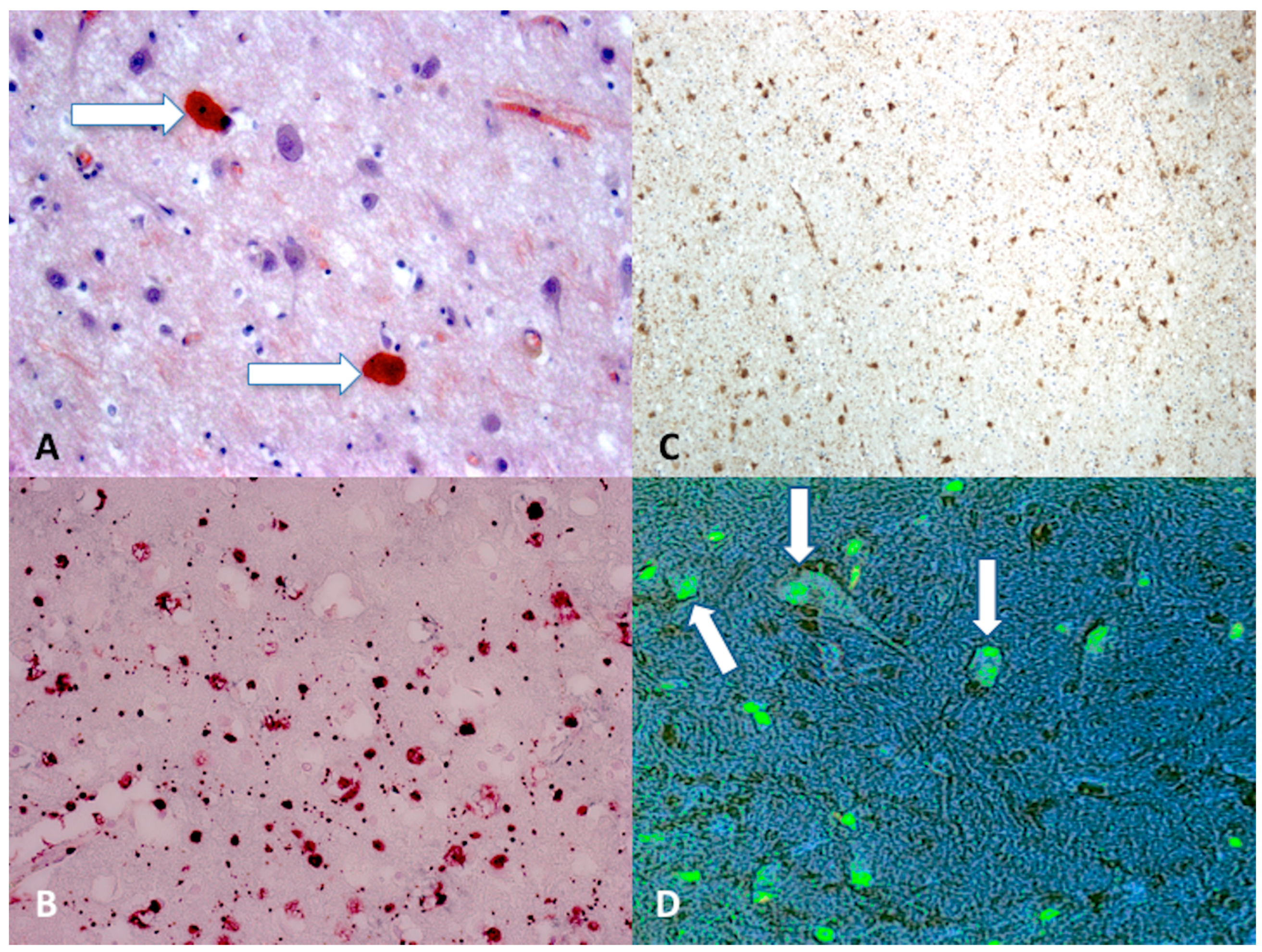
🔴 Red Neuron (Acute Ischemic Neuron) — VERY HIGH-YIELD
🔶 Liquefactive Necrosis (CNS Specific)
🔶 Selective Vulnerability (VERY IMPORTANT MCQ)
📊 Table: Selective Vulnerability
| Region |
Cells Affected |
| Hippocampus |
CA1 pyramidal neurons |
| Cerebellum |
Purkinje cells |
| Cerebral cortex |
Layers 3 & 5 neurons |
| Watershed zones |
Border areas (ACA–MCA) |
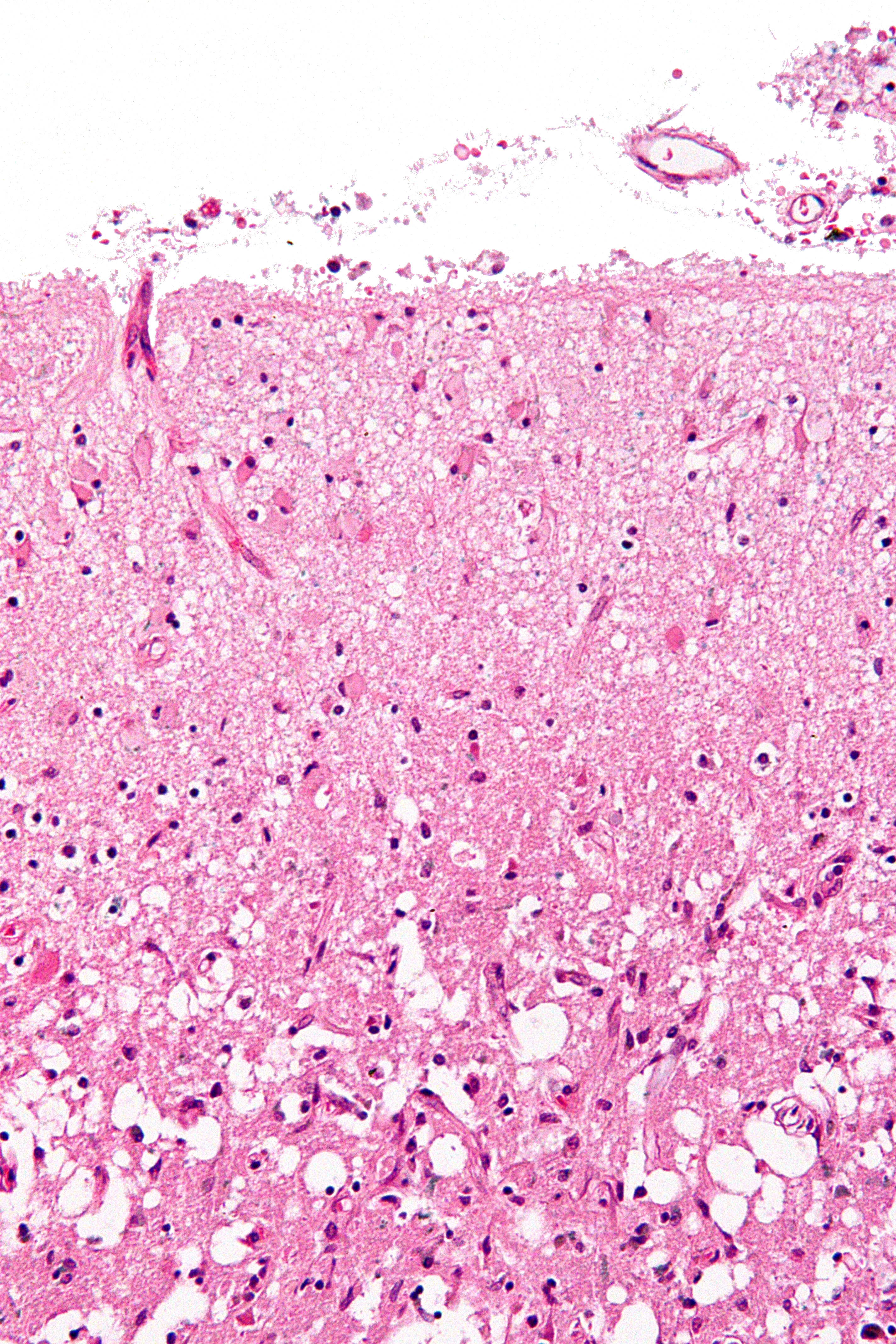
🔶 Astrocyte Response (Gliosis)
🔹 Functions
🔶 Oligodendrocyte Injury
🔶 Microglial Activation
-
CNS macrophages
-
Activated in:
🔹 Features
-
Proliferation
-
Formation of:
-
Microglial nodules
-
Neuronophagia
🔶 Axonal Injury
🔹 Changes
🔶 Regeneration vs Permanent Damage
📊 TABLES
📊 Table: CNS Cells vs Functions
| Cell Type |
Function |
| Neurons |
Signal transmission |
| Astrocytes |
Support, BBB, repair |
| Oligodendrocytes |
Myelination |
| Microglia |
Phagocytosis |
| Ependymal cells |
CSF lining |
📊 Table: Reversible vs Irreversible Neuronal Injury
| Feature |
Reversible |
Irreversible |
| Cell swelling |
Present |
Severe |
| Mitochondria |
Mild damage |
Severe damage |
| Nucleus |
Normal |
Pyknosis/karyorrhexis |
| Outcome |
Recovery possible |
Cell death |
📊 Table: Selective Vulnerability (HIGH-YIELD)
| Structure |
Vulnerability |
| Hippocampus (CA1) |
Most sensitive |
| Purkinje cells |
Highly sensitive |
| Cortex layers 3,5 |
Moderate |
| Watershed areas |
Hypotension sensitive |
🔬 SLIDES (EXAM FAVORITE)
🔬 Red Neuron

🔬 Gliosis
🔬 Microglial Nodules
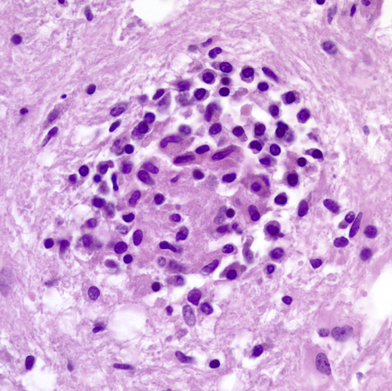

-
Clusters of microglia
-
Surround damaged neurons
-
Seen in viral infections
🧠 DIAGRAMS
🧠 Neuron Injury → Gliosis Pathway
Injury
↓
Neuronal damage
↓
Microglial activation
↓
Astrocyte proliferation
↓
Glial scar formation (gliosis)
🧠 CNS Cellular Response Diagram
Injury
↓
Neurons → necrosis
↓
Microglia → phagocytosis
↓
Astrocytes → gliosis
↓
Oligodendrocytes → demyelination
↓
Outcome:
-
Repair (gliosis)
-
Permanent deficit
🟢 FINAL HIGH-YIELD SUMMARY
-
Red neuron = marker of acute ischemia
-
Liquefactive necrosis = hallmark of brain infarction
-
Gliosis = main CNS repair mechanism
-
Selective vulnerability = important exam concept
-
Microglial nodules = seen in viral infections
-
CNS has minimal regenerative capacity
🧠 CENTRAL NERVOUS SYSTEM
🔶 EDEMA, HERNIATION, HYDROCEPHALUS
🔷 CEREBRAL EDEMA
🔹 Types
🔸 Vasogenic Edema
-
Most common type
-
Due to blood-brain barrier (BBB) disruption
-
Fluid accumulates in extracellular space
-
Predominantly affects white matter
🔸 Cytotoxic Edema
-
Due to cellular injury (ATP depletion)
-
Failure of Na⁺/K⁺ pump → intracellular swelling
-
Affects neurons, astrocytes, endothelial cells
-
Involves gray + white matter
🔸 Interstitial Edema
🔹 Causes
🔹 Pathogenesis
-
BBB disruption → vasogenic edema
-
Energy failure → cytotoxic edema
-
CSF obstruction → interstitial edema
-
↑ Intracranial pressure → ↓ cerebral perfusion → secondary injury
📊 TABLE
📊 Vasogenic vs Cytotoxic Edema
| Feature |
Vasogenic Edema |
Cytotoxic Edema |
| Mechanism |
BBB disruption |
Cellular injury |
| Fluid location |
Extracellular |
Intracellular |
| Area involved |
White matter |
Gray + white matter |
| Causes |
Tumor, inflammation |
Ischemia, hypoxia |
| Reversibility |
Potentially reversible |
Often irreversible |
🔬 SLIDES (EXAM FAVORITE)
🔬 Edematous Brain (Flattened Gyri)
🧠 DIAGRAM
🧠 Edema Pathogenesis Flowchart
Injury (tumor / ischemia / trauma)
↓
BBB disruption / ATP depletion
↓
Fluid accumulation
↓
Brain swelling
↓
↑ Intracranial pressure
↓
↓ Cerebral perfusion
↓
Secondary neuronal injury
🔷 HYDROCEPHALUS
🔹 Types
🔸 Non-communicating (Obstructive)
🔸 Communicating
🔸 Normal Pressure Hydrocephalus
🔹 Causes
-
Congenital malformations
-
Tumors
-
Infections (meningitis)
-
Hemorrhage
-
Aqueductal stenosis
🔹 Pathophysiology
-
CSF accumulation → ventricular dilation
-
Compression of brain parenchyma
-
Periventricular ischemia
-
In infants → head enlargement
-
In adults → increased intracranial pressure
📊 TABLE
📊 Types of Hydrocephalus
| Type |
Mechanism |
Example |
| Non-communicating |
Obstruction of CSF flow |
Aqueductal stenosis |
| Communicating |
Impaired absorption |
Meningitis |
| Normal pressure |
Chronic CSF imbalance |
Elderly patients |
🔬 SLIDES (EXAM FAVORITE)
🔬 Ventricular Dilation


🧠 DIAGRAM
🧠 CSF Circulation Pathway
Choroid plexus
↓
Lateral ventricles
↓
Foramen of Monro
↓
Third ventricle
↓
Aqueduct of Sylvius
↓
Fourth ventricle
↓
Foramina of Luschka & Magendie
↓
Subarachnoid space
↓
Arachnoid villi
↓
Venous sinuses
🟢 FINAL HIGH-YIELD SUMMARY
-
Vasogenic edema = BBB breakdown
-
Cytotoxic edema = cell injury (ATP failure)
-
Interstitial edema = CSF leakage
-
Flattened gyri = hallmark of cerebral edema
-
Hydrocephalus = ventricular dilation
-
Normal pressure hydrocephalus triad = gait + incontinence + dementia
-
CSF flow pathway = very important for exams
🔷 HERNIATION
🔹 Types
🔸 Subfalcine (Cingulate) Herniation
🔸 Transtentorial (Uncal) Herniation
🔹 Effects
-
Compression of CN III (oculomotor nerve) → ipsilateral dilated pupil
-
Compression of posterior cerebral artery (PCA) → occipital infarction
-
Compression of brainstem → altered consciousness
🔸 Tonsillar Herniation
🔹 Effects
-
Compression of medulla
-
Leads to:
🔹 Clinical Effects (General)
-
Increased intracranial pressure
-
Headache, vomiting
-
Altered consciousness
-
Pupillary changes
-
Focal neurological deficits
-
Brainstem compression → life-threatening
📊 TABLE
📊 Types of Herniation vs Features
| Type |
Structure Involved |
Structure Compressed |
Clinical Feature |
| Subfalcine |
Cingulate gyrus |
ACA |
Leg weakness |
| Transtentorial (Uncal) |
Medial temporal lobe |
CN III, PCA |
Dilated pupil, vision loss |
| Tonsillar |
Cerebellar tonsils |
Medulla |
Respiratory arrest |
🔬 SLIDES (EXAM FAVORITE)
🔬 Brain Compression Findings
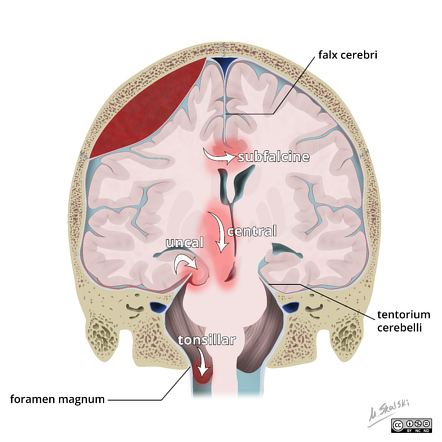

🧠 DIAGRAM
🧠 Brain Herniation Diagram
Mass lesion (tumor / hemorrhage / edema)
↓
Increased intracranial pressure
↓
Displacement of brain tissue
↓
Types:
-
Subfalcine → cingulate under falx
-
Transtentorial → uncus through tentorium
-
Tonsillar → cerebellum through foramen magnum
↓
Compression of vessels / nerves / brainstem
↓
Ischemia + neurological deficits
↓
Death (if untreated)
🟢 FINAL HIGH-YIELD SUMMARY
-
Herniation = displacement of brain due to increased ICP
-
Subfalcine → ACA compression → leg weakness
-
Uncal → CN III palsy → dilated pupil
-
Tonsillar → medullary compression → death
-
Midline shift = important radiological finding
-
Brainstem compression = most dangerous outcome
🔶 CEREBROVASCULAR DISEASES
🔷 HYPOXIA, ISCHEMIA, INFARCTION
🔹 Global Ischemia
🔹 Effects
🔹 Focal Ischemia
🔹 Effects
🔹 Infarction Types
🔸 Pale (Non-hemorrhagic) Infarct
🔸 Hemorrhagic Infarct
🔶 Liquefactive Necrosis
🔶 Selective Vulnerability (HIGH-YIELD)
🔹 Most Vulnerable
🔶 Watershed Infarcts (VERY IMPORTANT)
🔹 Causes
🔹 Features
📊 TABLES
📊 Global vs Focal Ischemia
| Feature |
Global Ischemia |
Focal Ischemia |
| Cause |
Hypotension / hypoxia |
Thrombosis / embolism |
| Distribution |
Diffuse |
Localized |
| Neuronal damage |
Selective vulnerability |
Territory-specific infarct |
| Example |
Cardiac arrest |
MCA infarct |
📊 Vulnerable Neurons
| Region |
Cells |
| Hippocampus |
CA1 neurons |
| Cerebellum |
Purkinje cells |
| Cortex |
Layers 3 & 5 |
| Watershed zones |
Border areas |
🔬 SLIDES (EXAM FAVORITE)
🔬 Red Neurons

🔬 Liquefactive Necrosis
🧠 DIAGRAM
🧠 Ischemia → Infarction Pathway
Vascular occlusion (thrombus / embolus)
↓
Reduced blood flow
↓
Hypoxia
↓
ATP depletion
↓
Ion pump failure (Na⁺/K⁺)
↓
Cellular swelling (cytotoxic edema)
↓
Calcium influx
↓
Enzyme activation
↓
Cell death
↓
Liquefactive necrosis
↓
Cyst formation
🟢 FINAL HIGH-YIELD SUMMARY
-
Global ischemia = diffuse brain injury
-
Focal ischemia = territorial infarct
-
Liquefactive necrosis = hallmark of CNS infarction
-
Red neurons = early ischemic change
-
Selective vulnerability = important exam concept
-
Watershed infarcts = hypotension-related injury
-
MCA territory = most commonly affected
🔷 INTRACRANIAL HEMORRHAGE
🔹 Epidural Hematoma
- Bleeding between skull and dura mater
- Cause:
- Trauma → middle meningeal artery rupture
- Classically associated with:
🔹 Features
- Lucid interval (temporary recovery after injury)
- Rapid neurological deterioration
🔹 Subdural Hematoma
- Bleeding between dura and arachnoid mater
- Cause:
- Common in:
- Elderly (brain atrophy)
- Infants
🔹 Features
- Slow progression
- Chronic hematoma possible
🔹 Subarachnoid Hemorrhage (SAH)
- Bleeding into subarachnoid space
🔹 Causes
- Berry aneurysm rupture (Circle of Willis)
- Trauma
🔹 Features
- Sudden severe headache:
- Neck stiffness
- Photophobia
🔹 Intraparenchymal Hemorrhage
- Bleeding within brain parenchyma
🔹 Causes
- Hypertension (most common)
- Charcot–Bouchard microaneurysms
- Amyloid angiopathy
🔹 Common Sites
- Basal ganglia
- Thalamus
- Pons
📊 TABLE
📊 Epidural vs Subdural vs Subarachnoid Hemorrhage
| Feature |
Epidural |
Subdural |
Subarachnoid |
| Location |
Skull–dura |
Dura–arachnoid |
Subarachnoid space |
| Vessel |
Middle meningeal artery |
Bridging veins |
Berry aneurysm |
| Onset |
Rapid |
Slow |
Sudden |
| Classic feature |
Lucid interval |
Gradual decline |
Thunderclap headache |
| CT finding |
Biconvex (lens-shaped) |
Crescent-shaped |
Diffuse blood in CSF |
🔬 SLIDES (EXAM FAVORITE)
🔬 Hemorrhage Sections


- Accumulation of blood
- Compression of adjacent brain tissue
- Disruption of normal architecture
🧠 DIAGRAM
🧠 Hemorrhage Location Diagram
Trauma / vascular rupture
↓
Locations:
- Epidural → skull–dura (arterial)
- Subdural → dura–arachnoid (venous)
- Subarachnoid → CSF space
- Intraparenchymal → brain tissue
↓
Mass effect
↓
↑ Intracranial pressure
↓
Brain compression
↓
Neurological deficits
🔷 OTHER VASCULAR DISEASES
🔹 Hypertensive Encephalopathy
- Due to severe uncontrolled hypertension
- Leads to:
- Cerebral edema
- Vascular damage
🔹 Features
- Headache
- Confusion
- Seizures
🔹 Lacunar Infarcts
- Small infarcts in deep brain structures
🔹 Common Sites
- Basal ganglia
- Thalamus
- Internal capsule
🔹 Cause
- Chronic hypertension → small vessel disease
🔹 Vascular Malformations
- Congenital abnormalities of vessels
🔹 Types
- Arteriovenous malformation (AVM)
- Cavernous malformation
📊 TABLE
📊 Small Vessel vs Large Vessel Disease
| Feature |
Small Vessel Disease |
Large Vessel Disease |
| Vessel size |
Small penetrating arteries |
Major cerebral arteries |
| Cause |
Hypertension |
Atherosclerosis |
| Lesions |
Lacunar infarcts |
Territorial infarcts |
| Common site |
Basal ganglia |
Cortex |
🔬 SLIDES (EXAM FAVORITE)
🔬 Lipohyalinosis


- Thickened vessel wall
- Hyaline deposition
- Luminal narrowing
🧠 DIAGRAM
🧠 Hypertension → Vessel Damage Pathway
Chronic hypertension
↓
Endothelial damage
↓
Hyaline deposition (lipohyalinosis)
↓
Vessel wall thickening
↓
Luminal narrowing
↓
Reduced blood flow
↓
Lacunar infarction / hemorrhage
🟢 FINAL HIGH-YIELD SUMMARY
- Epidural hematoma = middle meningeal artery + lucid interval
- Subdural hematoma = bridging vein rupture + slow onset
- Subarachnoid hemorrhage = berry aneurysm + sudden headache
- Intraparenchymal hemorrhage = hypertension-related
- Lacunar infarcts = small vessel disease (basal ganglia)
- Lipohyalinosis = hallmark of hypertensive vascular damage
- CT patterns = highly important for exams
🔶 CNS TRAUMA
🔷 TRAUMATIC PARENCHYMAL INJURIES
🔹 Concussion
- Mild traumatic brain injury
- Transient loss of neurological function
- No structural brain damage
🔹 Features
- Loss of consciousness (brief)
- Confusion
- Amnesia
🔹 Contusion
- Bruising of brain tissue due to trauma
- Occurs at:
- Site of impact (coup)
- Opposite side (contrecoup)
🔹 Common Sites
- Frontal lobes
- Temporal lobes
🔹 Features
- Hemorrhage
- Edema
- Tissue necrosis
🔹 Diffuse Axonal Injury (DAI)
- Severe brain injury due to shearing forces
- Caused by:
- Rapid acceleration–deceleration (e.g., road traffic accidents)
🔹 Pathology
- Axonal stretching and tearing
- Disruption of neuronal connections
🔹 Common Sites
- Corpus callosum
- Brainstem
- Gray-white matter junction
🔹 Clinical Features
- Immediate loss of consciousness
- Persistent coma
📊 TABLE
📊 Concussion vs Contusion vs Diffuse Axonal Injury
| Feature |
Concussion |
Contusion |
Diffuse Axonal Injury |
| Severity |
Mild |
Moderate |
Severe |
| Structural damage |
Absent |
Present |
Diffuse axonal damage |
| Mechanism |
Functional disturbance |
Direct impact |
Shearing forces |
| Loss of consciousness |
Brief |
Variable |
Immediate, prolonged |
| Outcome |
Reversible |
May progress |
Often poor prognosis |
🔬 SLIDES (EXAM FAVORITE)
🔬 Axonal Retraction Balls

- Swollen axonal ends
- Disconnected axons
- Marker of diffuse axonal injury
🧠 DIAGRAM
🧠 Diffuse Axonal Injury Mechanism
Rapid acceleration–deceleration
↓
Shearing forces
↓
Axonal stretching
↓
Axonal rupture
↓
Disconnection of neurons
↓
Diffuse brain dysfunction
↓
Coma
🔷 TRAUMATIC VASCULAR INJURY
🔹 Hemorrhage Types
- Epidural hemorrhage
- Subdural hemorrhage
- Subarachnoid hemorrhage
- Intraparenchymal hemorrhage
🔹 Secondary Ischemia
- Trauma → increased intracranial pressure
- ↓ Cerebral blood flow
- Leads to:
- Hypoxia
- Neuronal injury
- Infarction
🟢 FINAL HIGH-YIELD SUMMARY
- Concussion = transient functional disturbance
- Contusion = structural brain injury (coup–contrecoup)
- DAI = shearing injury → immediate coma
- Axonal retraction balls = hallmark of DAI
- Corpus callosum + brainstem = commonly affected
- Secondary ischemia = major cause of worsening injury
🔶 CONGENITAL MALFORMATIONS & PERINATAL INJURY
🔷 MALFORMATIONS
🔹 Neural Tube Defects
- Due to failure of neural tube closure (day 22–28 of gestation)
- Strong association with folate deficiency
🔹 Types
- Anencephaly → absence of brain and skull
- Spina bifida:
- Occulta
- Meningocele
- Myelomeningocele
🔹 Chiari Malformation
- Structural defect involving cerebellum and brainstem
🔹 Types
- Type I:
- Cerebellar tonsillar herniation
- Often asymptomatic
- Type II (Arnold–Chiari):
- Associated with myelomeningocele
- Brainstem involvement
🔹 Dandy–Walker Malformation
- Congenital malformation of posterior fossa
🔹 Features
- Enlarged posterior fossa
- Cystic dilation of 4th ventricle
- Agenesis of cerebellar vermis
🔹 Agenesis of Corpus Callosum
- Failure of development of corpus callosum
🔹 Features
- Cognitive impairment
- Seizures
- May be asymptomatic
📊 TABLE
📊 CNS Malformations Comparison
| Condition |
Defect |
Key Features |
| Neural tube defects |
Failure of tube closure |
Anencephaly, spina bifida |
| Chiari malformation |
Cerebellar herniation |
Brainstem compression |
| Dandy-Walker |
Posterior fossa defect |
Cystic dilation |
| Agenesis of corpus callosum |
Absent commissural fibers |
Cognitive deficits |
🔬 SLIDES (EXAM FAVORITE)
🔬 Neural Tube Defects


- Open neural tube
- Exposed neural tissue
- Structural deformity
🧠 DIAGRAM
🧠 Neural Tube Development
Neural plate formation
↓
Neural fold elevation
↓
Neural tube closure (day 22–28)
↓
Brain and spinal cord formation
Failure of closure
↓
Neural tube defects
🔷 PERINATAL BRAIN INJURY
🔹 Hypoxic Ischemic Encephalopathy (HIE)
- Due to perinatal asphyxia
🔹 Causes
- Placental insufficiency
- Birth complications
🔹 Features
- Diffuse brain injury
- Selective neuronal necrosis
🔹 Germinal Matrix Hemorrhage
- Occurs in premature infants
🔹 Pathogenesis
- Fragile vessels in germinal matrix
- Bleeding into ventricles
🔹 Periventricular Leukomalacia (PVL)
- White matter injury around ventricles
🔹 Causes
- Ischemia in premature infants
🔹 Features
- Necrosis of white matter
- Leads to cerebral palsy
📊 TABLE
📊 Term vs Preterm Brain Injury
| Feature |
Term Infants |
Preterm Infants |
| Type of injury |
Cortical injury |
Periventricular injury |
| Common lesion |
Hypoxic injury |
PVL |
| Hemorrhage |
Less common |
Germinal matrix hemorrhage |
| Outcome |
Cognitive deficits |
Motor deficits |
🔬 SLIDES (EXAM FAVORITE)
🔬 Periventricular Leukomalacia

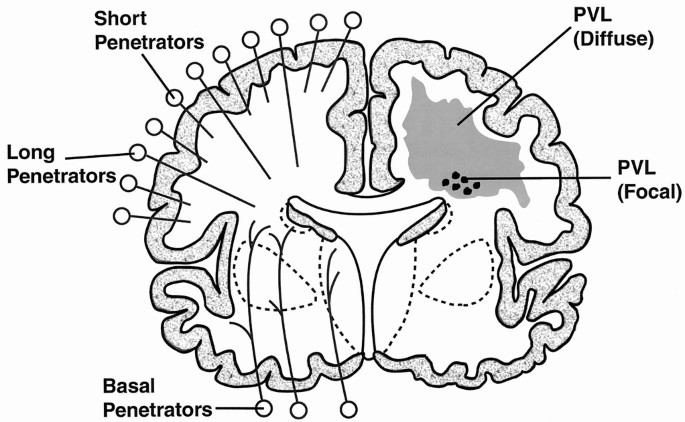
- Necrosis of periventricular white matter
- Cystic lesions
- Loss of normal structure
🧠 DIAGRAM
🧠 Hypoxia → Neonatal Brain Injury Pathway
Perinatal hypoxia
↓
Reduced cerebral blood flow
↓
ATP depletion
↓
Cellular injury
↓
Outcomes:
- Hypoxic ischemic encephalopathy
- Germinal matrix hemorrhage
- Periventricular leukomalacia
🟢 FINAL HIGH-YIELD SUMMARY
- Neural tube defects = failure of closure (day 22–28)
- Folate deficiency = major risk factor
- Chiari II = associated with myelomeningocele
- Dandy-Walker = cystic dilation of 4th ventricle
- PVL = most important lesion in premature infants
- Germinal matrix hemorrhage = fragile vessels
- HIE = global hypoxic injury in neonates
🔶 INFECTIONS OF NERVOUS SYSTEM
🔷 EPIDURAL & SUBDURAL INFECTIONS
🔹 Abscess Formation
- Localized collection of pus between skull and meninges
- Types:
- Epidural abscess
- Subdural abscess
🔹 Pathogenesis
- Spread from:
- Skull infection (osteomyelitis)
- Sinusitis
- Otitis media
🔹 Spread
- Direct extension from adjacent infection
- Hematogenous spread (rare)
- Rapid spread in subdural space due to lack of barriers
🔷 MENINGITIS
🔹 Acute Bacterial Meningitis
🔹 Common Organisms
- Neonates:
- E. coli
- Group B Streptococcus
- Adults:
- Streptococcus pneumoniae
- Neisseria meningitidis
🔹 Pathology
- Neutrophilic exudate in subarachnoid space
- Thick purulent material
🔹 Viral (Aseptic) Meningitis
🔹 Causes
- Enteroviruses (most common)
- HSV
🔹 Pathology
- Lymphocytic infiltrate
- Mild inflammation
🔹 Chronic Meningitis
🔹 Causes
- Tuberculosis
- Fungal infections (Cryptococcus)
🔹 Features
- Granulomatous inflammation
- Basal meningitis (TB)
📊 TABLE
📊 Bacterial vs Viral vs TB Meningitis
| Feature |
Bacterial |
Viral |
TB/Fungal |
| Onset |
Acute |
Subacute |
Chronic |
| Cells |
Neutrophils |
Lymphocytes |
Lymphocytes |
| CSF protein |
High |
Mild ↑ |
High |
| CSF glucose |
Low |
Normal |
Low |
| Severity |
Severe |
Mild |
Moderate |
🔬 SLIDES (EXAM FAVORITE)
🔬 Neutrophilic Exudate (Bacterial Meningitis)


- Dense neutrophilic infiltrate
- Purulent exudate
- Thickened meninges
🔬 Lymphocytic Infiltrate (Viral Meningitis)


- Lymphocyte predominance
- Mild inflammation
- No pus formation
🧠 DIAGRAM
🧠 Meningitis Pathogenesis
Pathogen entry (blood / direct spread)
↓
Crosses blood-brain barrier
↓
Multiplication in CSF
↓
Inflammatory response
↓
Cytokine release
↓
Leukocyte infiltration
↓
Meningeal inflammation
↓
↑ Intracranial pressure
↓
Neurological symptoms
🔷 PARENCHYMAL INFECTIONS
🔹 Brain Abscess
- Localized infection in brain parenchyma
🔹 Causes
- Bacterial infection
- Spread from:
- Sinusitis
- Otitis
- Endocarditis
🔹 Features
- Central necrosis
- Surrounding edema
- Capsule formation
🔹 Viral Encephalitis
🔸 HSV Encephalitis (VERY HIGH-YIELD)
- Most common cause of fatal sporadic encephalitis
- Affects:
🔹 Features
- Hemorrhagic necrosis
- Edema
- Altered consciousness
🔹 Toxoplasmosis
- Seen in immunocompromised patients
- Causes:
📊 TABLE
📊 Brain Abscess vs Encephalitis
| Feature |
Brain Abscess |
Encephalitis |
| Cause |
Bacterial |
Viral |
| Lesion |
Localized |
Diffuse |
| Pus |
Present |
Absent |
| Necrosis |
Central |
Diffuse |
| Edema |
Marked |
Moderate |
🔬 SLIDES (EXAM FAVORITE)
🔬 Microabscess


- Small collections of neutrophils
- Necrotic center
🔬 Viral Inclusion Bodies
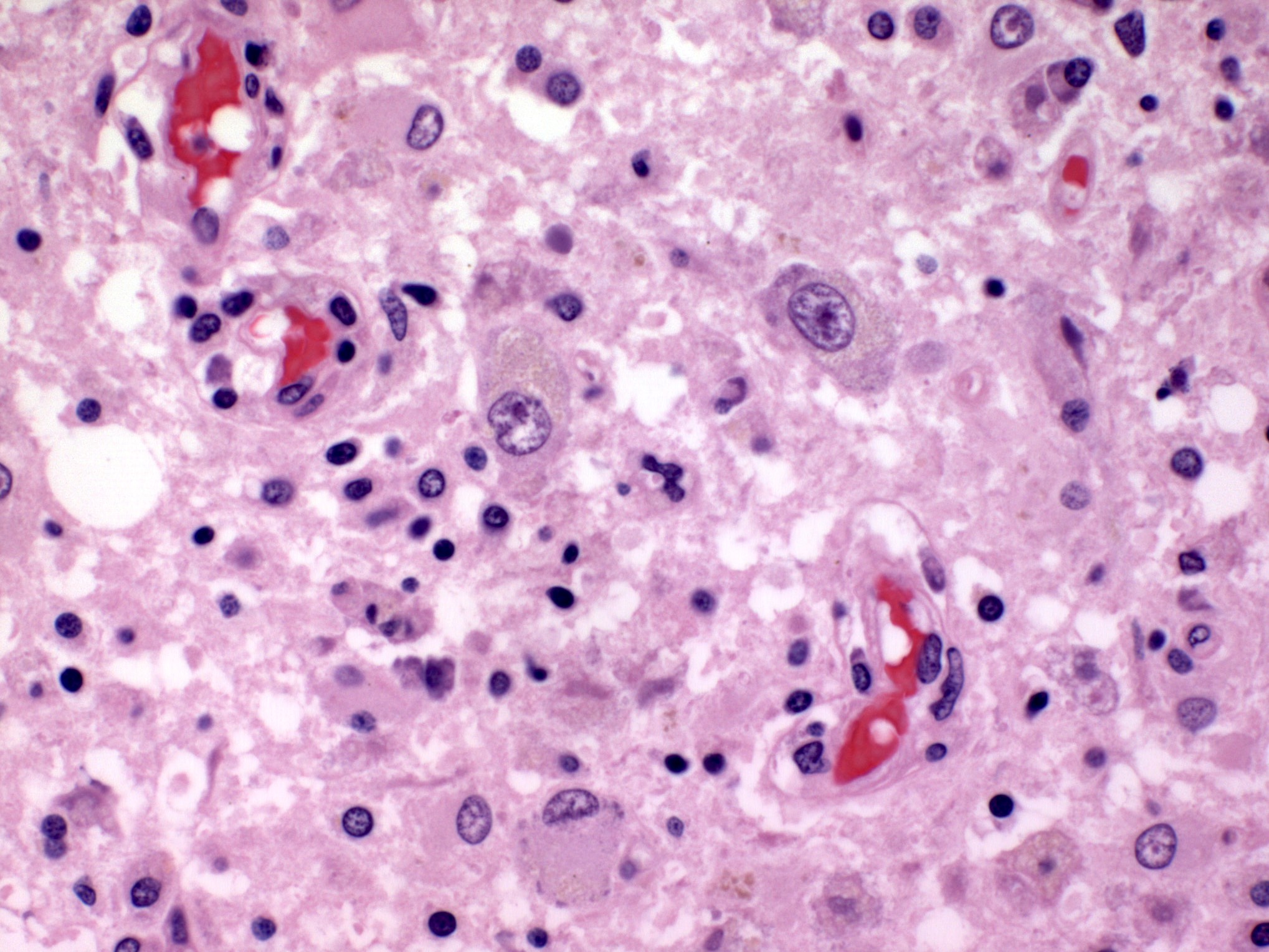

- Intranuclear inclusions
- Seen in viral infections (HSV)
🧠 DIAGRAM
🧠 Brain Abscess Formation
Infection (hematogenous / direct spread)
↓
Local inflammation
↓
Cerebritis (early stage)
↓
Necrosis
↓
Capsule formation
↓
Mature abscess
🟢 FINAL HIGH-YIELD SUMMARY
- Bacterial meningitis = neutrophilic exudate
- Viral meningitis = lymphocytic infiltrate
- TB meningitis = chronic granulomatous inflammation
- HSV encephalitis = temporal lobe + hemorrhagic necrosis
- Brain abscess = localized pus formation
- Toxoplasmosis = multiple lesions in immunocompromised
- Meningitis → increased ICP → neurological symptoms
🔷 CONGENITAL INFECTIONS (TORCH)
🔹 Subcontents
🔸 Cytomegalovirus (CMV)
- Most common congenital viral infection
- Affects:
🔹 Features
- Periventricular calcification
- Microcephaly
- Hearing loss
🔸 Toxoplasmosis
- Caused by Toxoplasma gondii
- Transmission:
🔹 Features
- Diffuse intracranial calcification
- Hydrocephalus
- Chorioretinitis
🔸 Rubella
- Congenital viral infection
🔹 Features
- Sensorineural deafness
- Cardiac defects
- Cataracts
📊 TABLE
📊 TORCH Infections Comparison
| Infection |
Key Feature |
Brain Finding |
| CMV |
Most common |
Periventricular calcification |
| Toxoplasmosis |
Severe infection |
Diffuse calcification |
| Rubella |
Congenital triad |
Mild CNS involvement |
🔷 PRION DISEASES
🔹 Creutzfeldt–Jakob Disease (CJD)
- Rapidly progressive neurodegenerative disease
- Caused by abnormal prion protein
🔹 Features
- Rapid dementia
- Myoclonus
- Fatal outcome
🔹 Pathogenesis
- Normal prion protein (PrPc) converts into abnormal form (PrPsc)
- Abnormal protein:
- Misfolded
- Protease resistant
- Leads to:
- Accumulation in brain
- Neuronal damage
📊 TABLE
📊 Types of Prion Diseases
| Type |
Example |
| Sporadic |
CJD |
| Familial |
Genetic mutation |
| Acquired |
Iatrogenic / variant CJD |
🔬 SLIDES (EXAM FAVORITE)
🔬 Spongiform Change

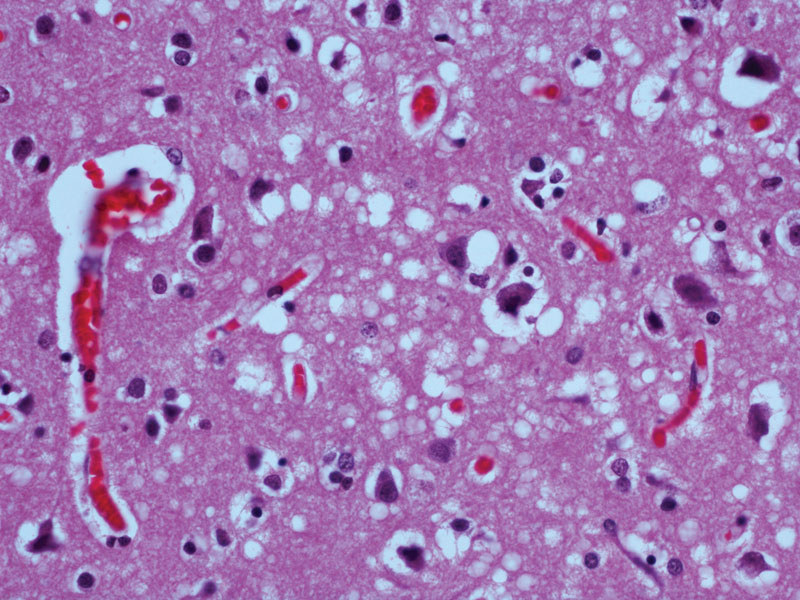
- Vacuolation of neuropil
- Sponge-like appearance
- Neuronal loss
- No inflammation
🧠 DIAGRAM
🧠 Prion Protein Conversion
Normal PrPc
↓
Misfolding
↓
Conversion to PrPsc
↓
Aggregation
↓
Neuronal damage
↓
Spongiform change
↓
Neurodegeneration
🟢 FINAL HIGH-YIELD SUMMARY
- CMV = periventricular calcification
- Toxoplasmosis = diffuse calcification
- Rubella = congenital triad
- Prion diseases = protein misfolding disorders
- PrPsc = abnormal infectious protein
- Spongiform change = hallmark of prion disease
- No inflammation in prion diseases
🔶 DEMYELINATING DISEASES
🔷 MULTIPLE SCLEROSIS
🔹 Autoimmune Pathogenesis
- Chronic immune-mediated demyelinating disease of CNS
- Involves:
- CD4+ T cells
- B cells (antibodies)
- Macrophages
🔹 Mechanism
- Trigger (environmental/genetic)
↓
Activation of autoreactive T cells
↓
Cross blood-brain barrier
↓
Recognition of myelin antigens
↓
Cytokine release
↓
Inflammation
↓
Demyelination
↓
Axonal damage
🔹 Plaques
- Characteristic lesion of MS
- Well-demarcated areas of:
🔹 Common Sites
- Periventricular white matter
- Optic nerves
- Brainstem
- Spinal cord
🔹 Clinical Features
- Relapsing-remitting course
- Visual disturbances:
- Motor weakness
- Sensory symptoms
- Ataxia
- Internuclear ophthalmoplegia
📊 TABLE
📊 Features of Multiple Sclerosis
| Feature |
Description |
| Pathogenesis |
Autoimmune demyelination |
| Lesion |
Plaques |
| Location |
Periventricular white matter |
| Course |
Relapsing-remitting |
| CSF finding |
Oligoclonal bands |
| Key symptom |
Optic neuritis |
🔬 SLIDES (EXAM FAVORITE)
🔬 Demyelinated Plaques
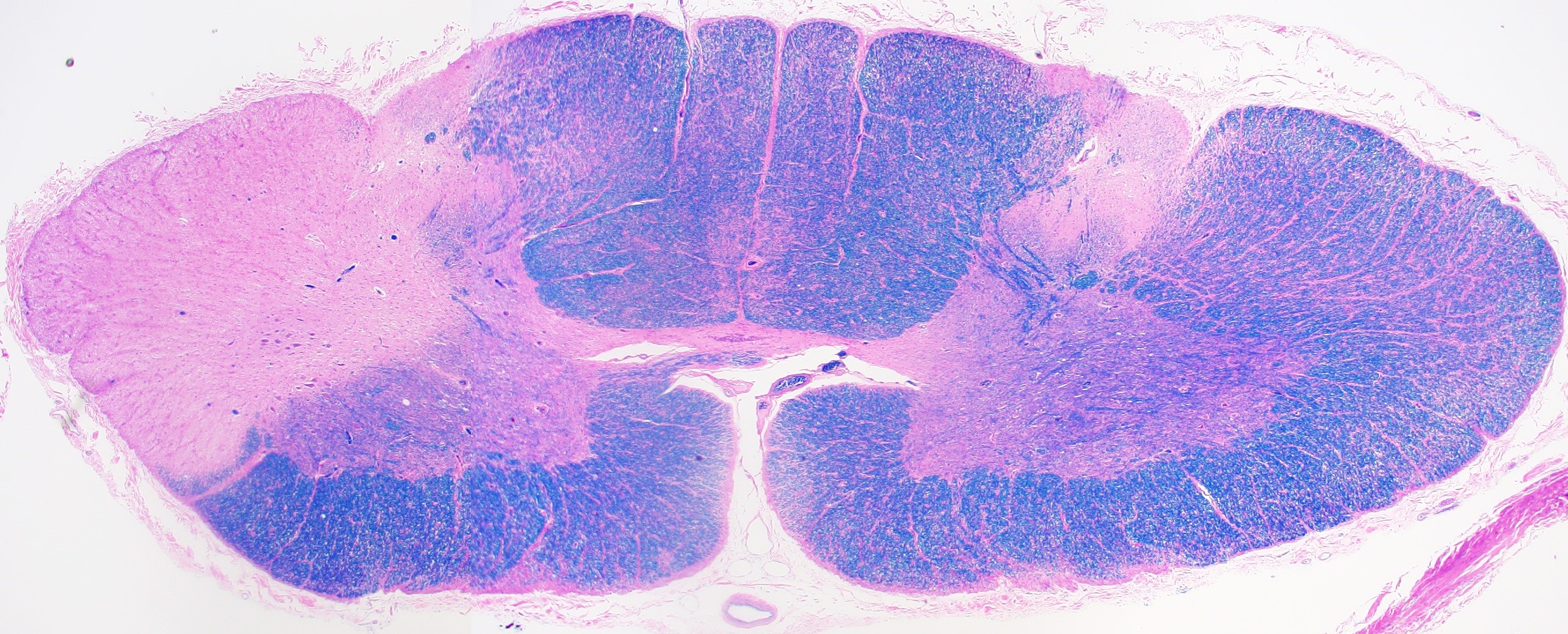

- Loss of myelin
- Relative preservation of axons (early)
- Inflammatory infiltrate
- Reactive gliosis
🧠 DIAGRAM
🧠 Autoimmune Demyelination Pathway
Trigger (genetic + environmental)
↓
T-cell activation
↓
Entry into CNS
↓
Myelin antigen recognition
↓
Cytokine release
↓
Inflammation
↓
Demyelination
↓
Plaque formation
↓
Neurological deficit
🔷 OTHER ACQUIRED DEMYELINATING DISEASES
🔹 Acute Disseminated Encephalomyelitis (ADEM)
- Acute inflammatory demyelinating disease
- Often follows:
- Viral infection
- Vaccination
🔹 Features
- Rapid onset
- Diffuse demyelination
- Monophasic course
🔹 Progressive Multifocal Leukoencephalopathy (PML)
- Caused by JC virus infection
- Occurs in:
- Immunocompromised patients (HIV, transplant)
🔹 Pathology
- Viral infection of oligodendrocytes
- Multifocal demyelination
🔹 Features
- Progressive neurological deficits
- No significant inflammation
🟢 FINAL HIGH-YIELD SUMMARY
- MS = autoimmune demyelinating disease
- Plaques = hallmark lesion
- Optic neuritis = common presentation
- Oligoclonal bands = diagnostic clue
- ADEM = post-infectious, monophasic
- PML = JC virus infection in immunocompromised
- Demyelination = central feature of all these diseases
🔷 LEUKODYSTROPHIES
🔹 Metachromatic Leukodystrophy
- Caused by arylsulfatase A deficiency
- Leads to accumulation of sulfatides
🔹 Pathology
- Progressive demyelination of CNS and peripheral nerves
🔹 Features
- Motor dysfunction
- Peripheral neuropathy
- Cognitive decline
🔹 Krabbe Disease
- Caused by galactocerebrosidase deficiency
- Leads to accumulation of psychosine (toxic to oligodendrocytes)
🔹 Pathology
- Severe demyelination
- Presence of globoid cells
🔹 Features
- Developmental delay
- Optic atrophy
- Peripheral neuropathy
📊 TABLE
📊 Leukodystrophies Classification
| Disease |
Enzyme Defect |
Accumulated Substance |
Key Feature |
| Metachromatic leukodystrophy |
Arylsulfatase A |
Sulfatides |
Peripheral neuropathy |
| Krabbe disease |
Galactocerebrosidase |
Psychosine |
Globoid cells |
🔶 METABOLIC & TOXIC DISORDERS
🔷 NUTRITIONAL DISEASES
🔹 Wernicke Encephalopathy
- Due to vitamin B1 (thiamine) deficiency
- Common in:
🔹 Triad
- Confusion
- Ophthalmoplegia
- Ataxia
🔹 Pathology
- Lesions in:
- Mammillary bodies
- Periventricular regions
🔹 Vitamin B12 Deficiency
- Leads to:
- Subacute combined degeneration
🔹 Pathology
- Demyelination of:
- Posterior columns
- Lateral corticospinal tracts
🔹 Features
- Paresthesia
- Ataxia
- Weakness
📊 TABLE
📊 Nutritional CNS Disorders
| Disorder |
Deficiency |
Key Features |
| Wernicke encephalopathy |
Vitamin B1 |
Confusion, ophthalmoplegia, ataxia |
| B12 deficiency |
Vitamin B12 |
Posterior column degeneration |
🔷 METABOLIC DISORDERS
🔹 Hepatic Encephalopathy
- Due to liver failure → ammonia accumulation
🔹 Pathology
- Alzheimer type II astrocytes
🔹 Features
- Altered mental status
- Asterixis
- Coma (severe cases)
🔹 Uremia
🔹 Features
- Encephalopathy
- Cognitive dysfunction
- Seizures
🔹 Central Pontine Myelinolysis
- Caused by rapid correction of hyponatremia
🔹 Pathology
🔹 Features
- Quadriplegia
- Dysphagia
- Locked-in syndrome (severe)
🟢 FINAL HIGH-YIELD SUMMARY
- Leukodystrophies = inherited demyelinating disorders
- Metachromatic leukodystrophy = sulfatide accumulation
- Krabbe disease = psychosine toxicity + globoid cells
- Wernicke encephalopathy = B1 deficiency triad
- B12 deficiency = posterior column demyelination
- Hepatic encephalopathy = ammonia toxicity + astrocyte changes
- Central pontine myelinolysis = rapid Na correction → pontine demyelination
🔷 TOXIC DISORDERS
🔹 Alcohol
- Chronic alcohol use → direct neurotoxicity + nutritional deficiency
🔹 Effects
- Cerebral atrophy
- Wernicke encephalopathy (B1 deficiency)
- Korsakoff syndrome:
- Memory loss
- Confabulation
🔹 Heavy Metals
🔸 Lead
- Interferes with neurotransmission
- Causes:
- Cognitive impairment
- Encephalopathy (children)
🔸 Mercury
- Causes:
- Sensory neuropathy
- Cerebellar dysfunction
🧠 DIAGRAM
🧠 Ammonia Toxicity Pathway
Liver failure
↓
Ammonia accumulation
↓
Crosses blood-brain barrier
↓
Astrocyte uptake
↓
Glutamine formation
↓
Osmotic swelling of astrocytes
↓
Cerebral edema
↓
Neuronal dysfunction
🔶 NEURODEGENERATIVE DISEASES
🔷 ALZHEIMER DISEASE
🔹 Pathogenesis
🔸 Amyloid β Deposition
- Derived from amyloid precursor protein (APP)
- Forms:
🔸 Tau Pathology
- Hyperphosphorylated tau protein
- Forms:
🔸 Genetics
- APP gene mutation (chromosome 21)
- Presenilin 1, Presenilin 2 mutations
- ApoE4 allele:
🔹 Morphology
- Diffuse cortical atrophy
- Widened sulci
- Enlarged ventricles
🔹 Microscopic Features
- Amyloid plaques
- Neurofibrillary tangles
- Neuronal loss
📊 TABLE
📊 Alzheimer Disease vs Normal Aging
| Feature |
Alzheimer Disease |
Normal Aging |
| Memory |
Severe impairment |
Mild decline |
| Brain size |
Marked atrophy |
Minimal change |
| Plaques |
Present |
Minimal |
| Tangles |
Prominent |
Rare |
| Progression |
Progressive |
Stable |
🔬 SLIDES (EXAM FAVORITE)
🔬 Amyloid Plaques


- Extracellular deposits
- β-amyloid protein
- Surrounded by dystrophic neurites
🔬 Neurofibrillary Tangles
- Intracellular aggregates
- Hyperphosphorylated tau
- Flame-shaped structures
🧠 DIAGRAM
🧠 Amyloid Cascade Hypothesis
APP
↓
Abnormal cleavage
↓
β-amyloid accumulation
↓
Plaque formation
↓
Tau phosphorylation
↓
Neurofibrillary tangles
↓
Neuronal dysfunction
↓
Neuronal death
↓
Dementia
🟢 FINAL HIGH-YIELD SUMMARY
- Alcohol = neurotoxicity + B1 deficiency
- Heavy metals = neuronal damage (lead, mercury)
- Ammonia toxicity = astrocyte swelling → cerebral edema
- Alzheimer = most common dementia
- Amyloid plaques = extracellular
- Neurofibrillary tangles = intracellular
- ApoE4 = major genetic risk factor
- Cortical atrophy = key gross feature
🔷 FRONTOTEMPORAL LOBAR DEGENERATION (FTLD)
🔹 Subcontents
🔸 Pathology
- Degeneration of frontal and temporal lobes
- Associated protein abnormalities:
- Tau protein (FTLD-tau)
- TDP-43 protein (FTLD-TDP)
🔹 Features
- Early behavioral changes
- Personality alteration
- Disinhibition
- Language disturbances (aphasia)
🔹 Morphology
- Focal cortical atrophy:
- Frontal lobe
- Temporal lobe
🔷 PARKINSON DISEASE
🔹 Subcontents
🔸 Substantia Nigra Degeneration
- Loss of dopaminergic neurons in substantia nigra (pars compacta)
- Leads to:
🔸 Dopamine Depletion
- Imbalance in basal ganglia circuits
- Results in:
🔸 Lewy Bodies
- Intracytoplasmic inclusions
- Composed of:
🔹 Clinical Features
- Resting tremor
- Rigidity
- Bradykinesia
- Postural instability
📊 TABLE
📊 Parkinson Disease vs Other Movement Disorders
| Feature |
Parkinson Disease |
Huntington Disease |
| Movement |
Hypokinetic |
Hyperkinetic |
| Neurotransmitter |
Dopamine ↓ |
GABA ↓ |
| Main site |
Substantia nigra |
Caudate nucleus |
| Tremor |
Present |
Absent |
| Onset |
Late |
Earlier |
🔬 SLIDES (EXAM FAVORITE)
🔬 Lewy Bodies

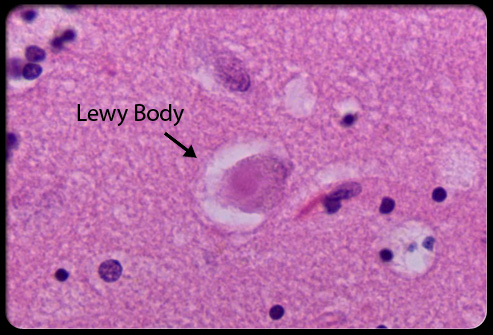
- Eosinophilic cytoplasmic inclusions
- Contain α-synuclein
- Seen in neurons of substantia nigra
🧠 DIAGRAM
🧠 Basal Ganglia Pathway
Substantia nigra degeneration
↓
Dopamine ↓
↓
↓ Stimulation of direct pathway
↑ Activity of indirect pathway
↓
Increased inhibitory output from basal ganglia
↓
Reduced thalamic stimulation
↓
Reduced motor cortex activity
↓
Bradykinesia and rigidity
🟢 FINAL HIGH-YIELD SUMMARY
- FTLD = frontal + temporal lobe degeneration
- Tau and TDP-43 = key proteins
- Parkinson = substantia nigra degeneration
- Dopamine ↓ = core mechanism
- Lewy bodies = α-synuclein inclusions
- Basal ganglia imbalance = movement disorder
- Parkinson = hypokinetic disorder
🔷 HUNTINGTON DISEASE
🔹 Subcontents
🔸 CAG Repeat Expansion
- Autosomal dominant disorder
- Mutation in HTT gene (chromosome 4)
- CAG trinucleotide repeat expansion
- Shows anticipation (earlier onset in successive generations)
🔸 Caudate Nucleus Atrophy
- Marked degeneration of:
- Caudate nucleus
- Putamen (to lesser extent)
- Leads to:
- Enlarged lateral ventricles
🔸 Neurotransmitter Changes
- GABA ↓ (inhibitory neurotransmitter)
- Also ↓ acetylcholine
🔹 Clinical Features
- Chorea (involuntary movements)
- Behavioral changes
- Dementia
📊 TABLE
📊 Trinucleotide Repeat Disorders
| Disease |
Repeat |
Gene |
Feature |
| Huntington disease |
CAG |
HTT |
Chorea |
| Fragile X |
CGG |
FMR1 |
Intellectual disability |
| Myotonic dystrophy |
CTG |
DMPK |
Muscle weakness |
🔷 SPINOCEREBELLAR ATAXIAS
🔹 Subcontents
- Group of genetic neurodegenerative disorders
- Characterized by:
- Progressive cerebellar degeneration
🔹 Features
- Ataxia
- Coordination defects
- Gait disturbances
🔷 AMYOTROPHIC LATERAL SCLEROSIS (ALS)
🔹 Subcontents
🔸 Motor Neuron Degeneration
- Degeneration of:
- Upper motor neurons
- Lower motor neurons
🔹 Features
- Muscle weakness
- Atrophy
- Fasciculations
- No sensory loss
🔶 CNS TUMORS
🔷 GLIOMAS
🔹 Astrocytoma
- Tumor of astrocytes
- Range from:
🔹 Glioblastoma
- Most aggressive primary brain tumor
🔹 Types
- Primary glioblastoma
- Secondary glioblastoma
- Progression from lower-grade astrocytoma
- p53 mutation
🔹 Oligodendroglioma
- Tumor of oligodendrocytes
🔹 Features
- 1p/19q co-deletion (diagnostic and prognostic)
🔹 Ependymoma
🔹 Features
- Common in children
- Located in:
📊 TABLE
📊 Glioma Classification
| Tumor |
Cell Type |
Key Feature |
| Astrocytoma |
Astrocytes |
Variable grade |
| Glioblastoma |
Astrocytes |
Highly aggressive |
| Oligodendroglioma |
Oligodendrocytes |
1p/19q deletion |
| Ependymoma |
Ependymal cells |
Ventricular tumor |
🔬 SLIDES (EXAM FAVORITE)
🔬 Pseudopalisading Necrosis (Glioblastoma)


- Tumor cells arranged around necrotic areas
- Highly characteristic of glioblastoma
🔬 Chicken-Wire Vessels (Oligodendroglioma)
- Delicate branching capillaries
- “Fried egg” cells
- Characteristic vascular pattern
🧠 DIAGRAM
🧠 Tumor Progression Pathway
Normal glial cell
↓
Genetic mutations
↓
Cell cycle dysregulation
↓
Uncontrolled proliferation
↓
Low-grade tumor
↓
Accumulation of mutations
↓
High-grade tumor (glioblastoma)
↓
Invasion and necrosis
🟢 FINAL HIGH-YIELD SUMMARY
- Huntington = CAG repeat + caudate atrophy + GABA ↓
- Spinocerebellar ataxia = cerebellar degeneration
- ALS = motor neuron disease (no sensory loss)
- Glioblastoma = most aggressive tumor
- EGFR (primary) and p53 (secondary) pathways
- Oligodendroglioma = 1p/19q deletion
- Ependymoma = 4th ventricle tumor in children
- Pseudopalisading necrosis = hallmark of glioblastoma
🔷 NEURONAL TUMORS
🔹 Ganglioglioma
- Tumor composed of:
- Neoplastic ganglion cells + glial cells
🔹 Features
- Usually low-grade tumor
- Common in:
🔹 Clinical
- Often presents with:
- Seizures (young patients)
🔷 EMBRYONAL TUMORS
🔹 Medulloblastoma
- Highly malignant embryonal tumor
- Common in:
🔹 Location
- Cerebellum (midline vermis)
🔹 Features
- Rapid growth
- Tendency to spread via CSF (drop metastasis)
🔬 SLIDES (EXAM FAVORITE)
🔬 Small Round Blue Cells
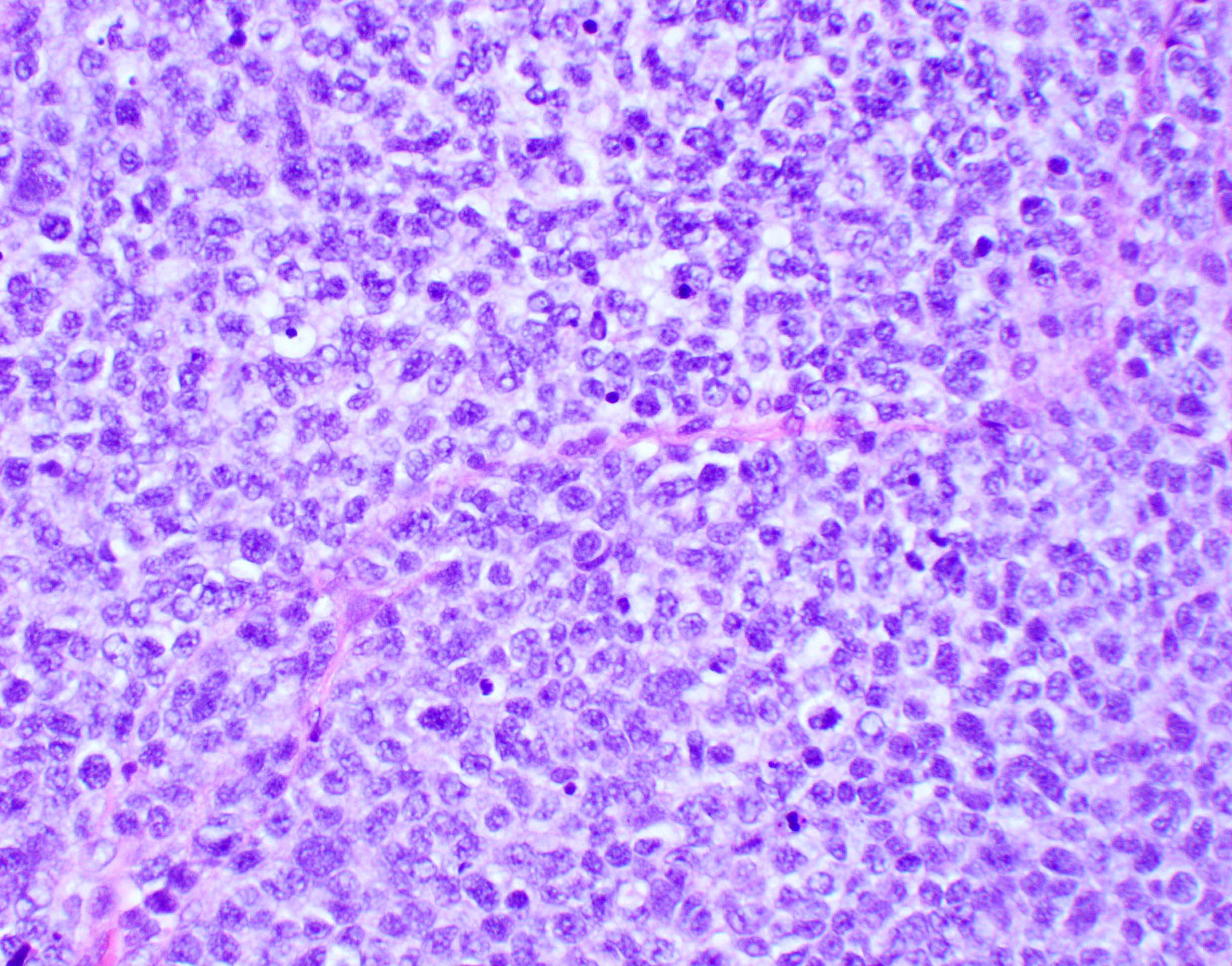
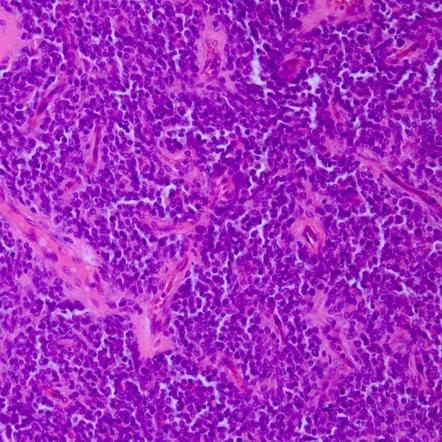
- Densely packed cells
- Hyperchromatic nuclei
- High nuclear-to-cytoplasmic ratio
🔬 Homer–Wright Rosettes

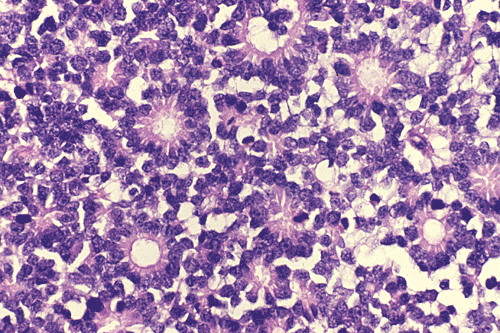
- Tumor cells arranged around central fibrillary space
- No true lumen
- Characteristic of medulloblastoma
🔷 OTHER PARENCHYMAL TUMORS
🔹 Examples
- Pineal tumors
- Choroid plexus tumors
🔷 MENINGIOMAS
🔹 Subcontents
- Tumor arising from arachnoid cap cells
- Usually benign
🔹 Key Features
- Dural attachment
- Associated with:
- Estrogen receptors (more common in females)
🔹 Morphology
- Whorled pattern of cells
- Presence of:
- Psammoma bodies (calcified structures)
🔬 SLIDES (EXAM FAVORITE)
🔬 Psammoma Bodies


- Concentric calcifications
- Seen in meningioma
🔷 METASTATIC TUMORS
🔹 Subcontents
- Most common brain tumors overall
🔹 Spread
🔹 Common Site
- Gray-white matter junction
🔹 Common Primary Tumors
🧠 DIAGRAM
🧠 Metastasis Pathway
Primary tumor
↓
Local invasion
↓
Intravasation
↓
Circulation
↓
Extravasation (brain)
↓
Tumor deposition at gray-white junction
↓
Metastatic tumor formation
🔷 FAMILIAL TUMOR SYNDROMES
🔹 Neurofibromatosis Type 1 (NF1)
- Mutation in NF1 gene
- Loss of neurofibromin
🔹 Features
- Neurofibromas
- Café-au-lait spots
- Optic glioma
🔹 Neurofibromatosis Type 2 (NF2)
- Mutation in NF2 gene
- Loss of merlin protein
🔹 Features
- Bilateral vestibular schwannomas
- Meningiomas
🔹 Tuberous Sclerosis
- Mutation in TSC1/TSC2 genes
🔹 Features
- Cortical tubers
- Subependymal giant cell astrocytoma
- Skin lesions
📊 TABLE
📊 Tumor Syndromes
| Syndrome |
Gene |
Key Tumors |
| NF1 |
NF1 |
Neurofibroma, glioma |
| NF2 |
NF2 |
Schwannoma, meningioma |
| Tuberous sclerosis |
TSC1/TSC2 |
Astrocytoma |
🧠 DIAGRAMS (VERY HIGH-YIELD)
🧠 NF1 Pathway
NF1 mutation
↓
Loss of neurofibromin
↓
RAS pathway activation
↓
Increased cell proliferation
↓
Tumor formation
🧠 NF2 Pathway
NF2 mutation
↓
Loss of merlin
↓
Loss of growth suppression
↓
Schwann cell proliferation
↓
Schwannoma formation
↓
Bilateral vestibular tumors
🧠 Tumor Genetics Pathway
Genetic mutation
↓
Oncogene activation
↓
Tumor suppressor loss
↓
Cell cycle dysregulation
↓
Uncontrolled proliferation
↓
Tumor growth
🟢 FINAL HIGH-YIELD SUMMARY
- Ganglioglioma = mixed neuronal + glial tumor
- Medulloblastoma = cerebellar tumor in children + CSF spread
- Homer-Wright rosettes = key histological feature
- Meningioma = dural-based tumor with psammoma bodies
- Metastasis = most common brain tumors (gray-white junction)
- NF1 = neurofibromin loss
- NF2 = merlin loss + bilateral schwannomas
- Tuberous sclerosis = cortical tubers + astrocytoma
Ready to study offline?
Get the full PDF version of this chapter.





























